The study intends to determine how short-term, high-intensity statin therapy affects the condition of blood vessels and how it might help prevent severe coronary artery disease.
 “This study is the result of more than a year of persistent preparatory work, which led to the granting of a European-level ethical approval. The research was launched at the initiative of leading experts at Semmelweis University, with funding from the OTKA grant, and is being carried out in collaboration with renowned domestic and international partners such as HeartFlow Ltd. (USA) and Charité University (Berlin, Germany). The drug being tested is a widely used cholesterol-lowering pill, which is repackaged by the University Pharmacy Department of Pharmacy Administration for the patients participating in the study, whom we are recruiting from several of the university’s clinics, including the Városmajor Heart and Vascular Center and the Department of Internal Medicine and Oncology. We are delighted that several hospitals outside Budapest have also joined the trial,” said Dr. Pál Maurovich Horvat, Director of the Medical Imaging Center at Semmelweis University and Lead Researcher of the clinical trial.
“This study is the result of more than a year of persistent preparatory work, which led to the granting of a European-level ethical approval. The research was launched at the initiative of leading experts at Semmelweis University, with funding from the OTKA grant, and is being carried out in collaboration with renowned domestic and international partners such as HeartFlow Ltd. (USA) and Charité University (Berlin, Germany). The drug being tested is a widely used cholesterol-lowering pill, which is repackaged by the University Pharmacy Department of Pharmacy Administration for the patients participating in the study, whom we are recruiting from several of the university’s clinics, including the Városmajor Heart and Vascular Center and the Department of Internal Medicine and Oncology. We are delighted that several hospitals outside Budapest have also joined the trial,” said Dr. Pál Maurovich Horvat, Director of the Medical Imaging Center at Semmelweis University and Lead Researcher of the clinical trial.
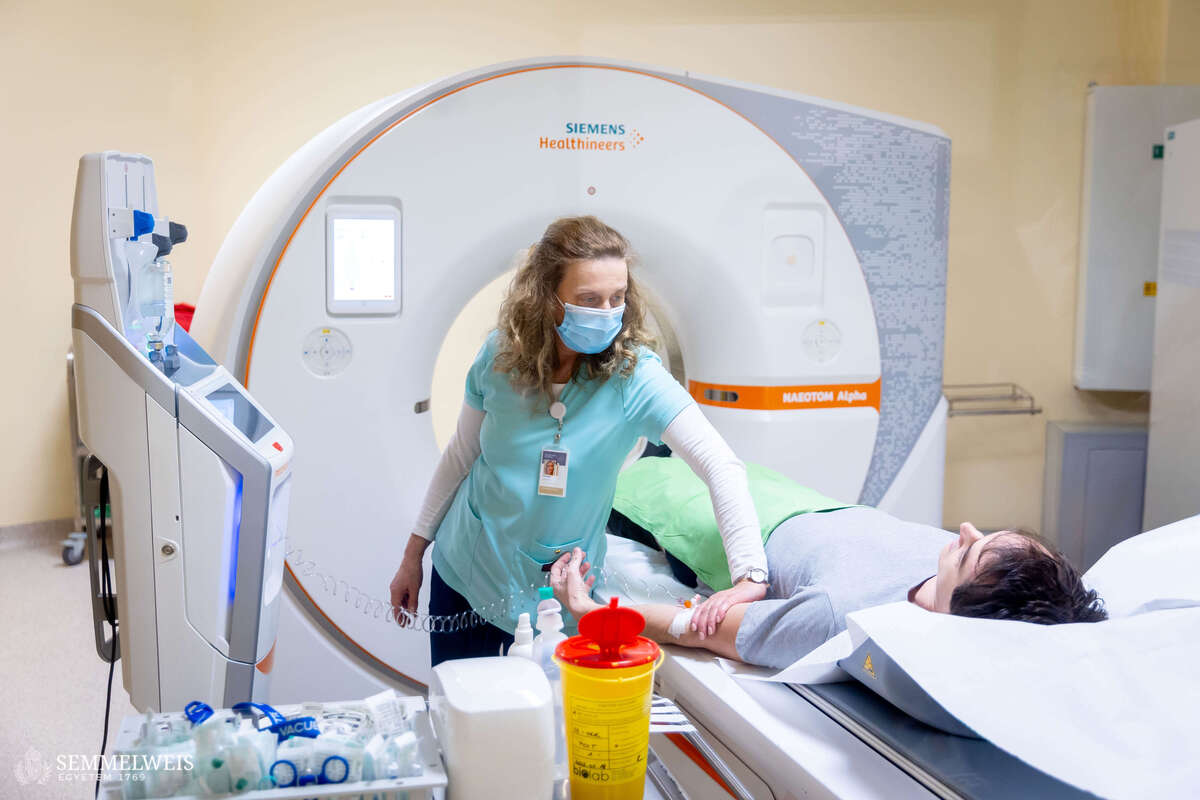
 “A patient who visits a cardiologist with chest pain and is diagnosed with high cholesterol will usually leave the consultation with a prescription for a statin-based cholesterol-lowering medication that they are expected to take for the rest of their lives. In most cases, the patient will take the medication diligently for a while but then stop after a few months or a year. Either because they forget, or because they think they’ve been taking it ‘long enough.’ In fact, no one has yet investigated how long it takes for statin to take effect. We know that it lowers the blood cholesterol levels and prevents further plaque buildup, but we do not know whether the medication starts working within three months or whether plaque stabilization begins within that timeframe. We are seeking answers to these questions using our state-of-the-art photon-counting CT scanner,” said Dr. Lili Száraz, one of the study’s coordinators, summarizing the essence of the clinical trial.
“A patient who visits a cardiologist with chest pain and is diagnosed with high cholesterol will usually leave the consultation with a prescription for a statin-based cholesterol-lowering medication that they are expected to take for the rest of their lives. In most cases, the patient will take the medication diligently for a while but then stop after a few months or a year. Either because they forget, or because they think they’ve been taking it ‘long enough.’ In fact, no one has yet investigated how long it takes for statin to take effect. We know that it lowers the blood cholesterol levels and prevents further plaque buildup, but we do not know whether the medication starts working within three months or whether plaque stabilization begins within that timeframe. We are seeking answers to these questions using our state-of-the-art photon-counting CT scanner,” said Dr. Lili Száraz, one of the study’s coordinators, summarizing the essence of the clinical trial.
The clinical coordinators of the study, Dr. Barnabás Baksa and Dr. Réka Sebestyén-Dósa, explained that while the entire study spanned two years, the short-term effects of statin therapy were to be evaluated over a three-month period.
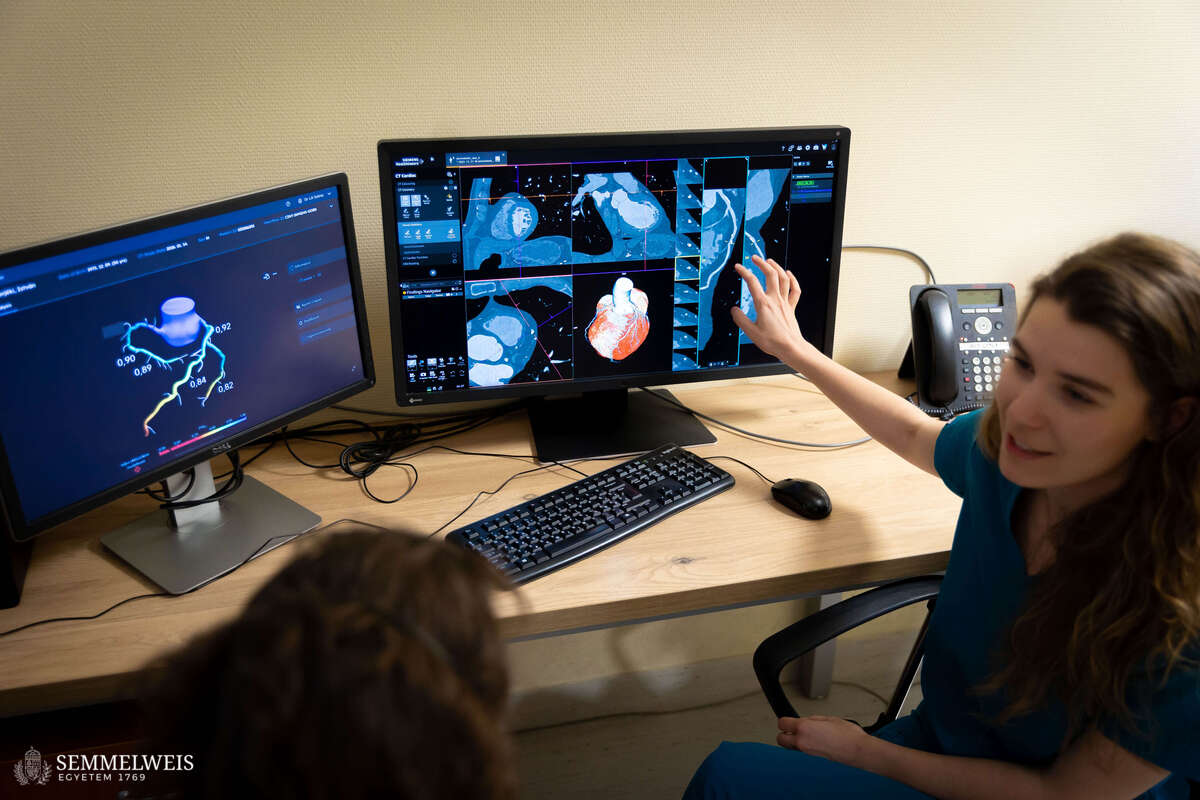
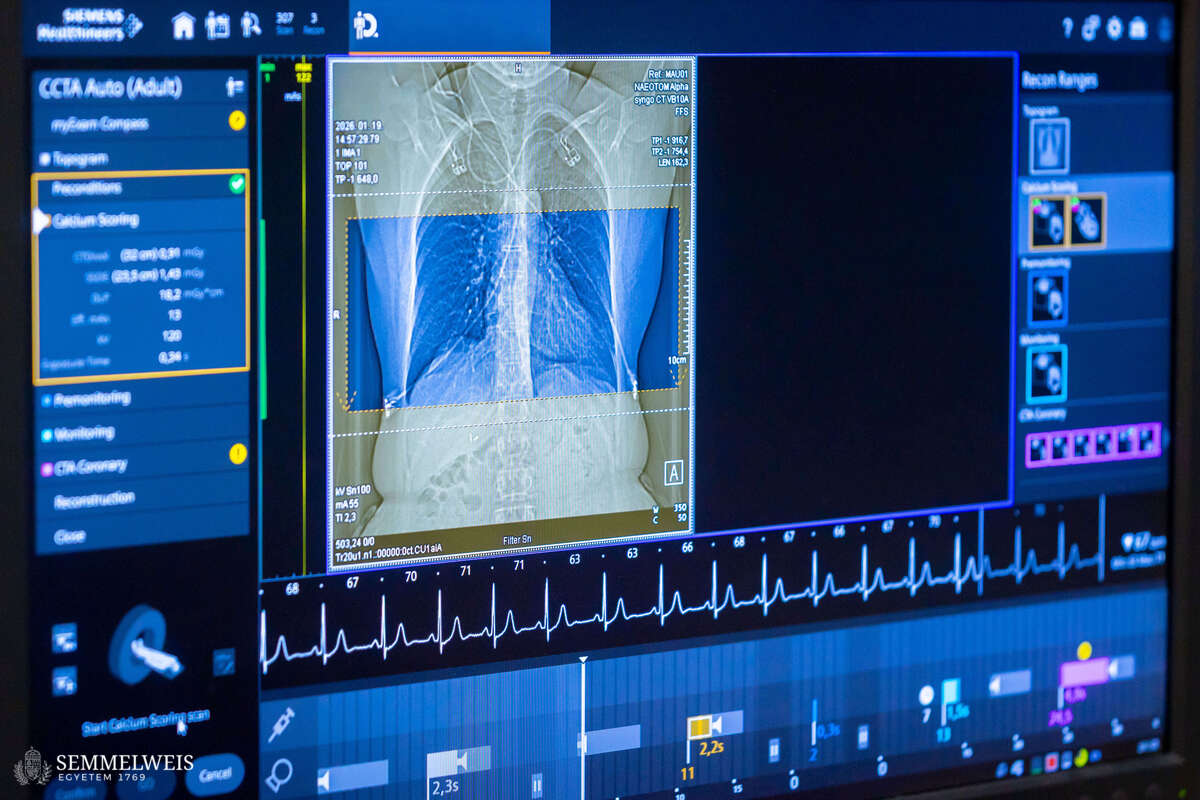
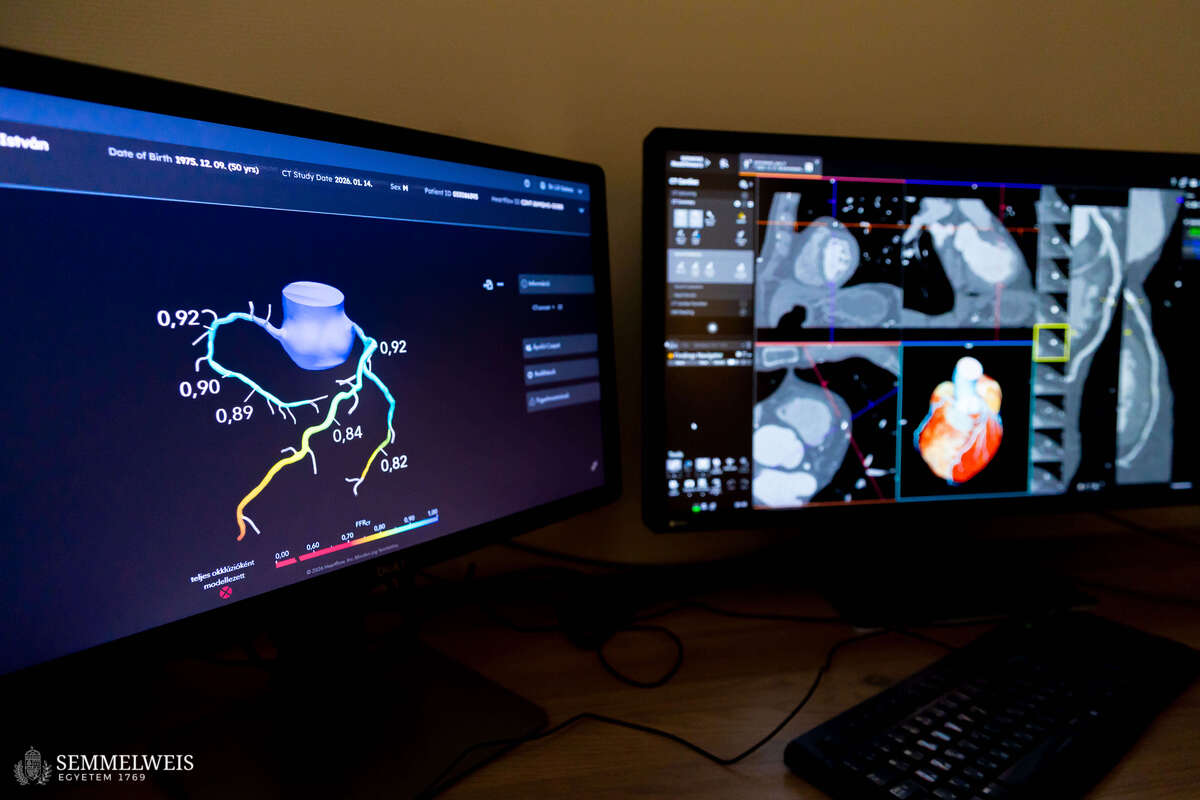
“In practice, this means that when a patient arrives at the Medical Imaging Center with chest complaints and is recommended a cardiac CT scan by a cardiologist, we first perform the scan. If coronary plaque or buildup on the walls of the blood vessels is confirmed, we discuss the possible next steps with the patient immediately afterwards. If, having received the detailed information, the patient wishes to participate in the study, we will analyze their CT scans in detail, including performing a coronary flow simulation. This will allow us to rule out the need for cardiac catheterization. If such a procedure is required, the patient will not be eligible to participate in the clinical trial. In addition to the cardiac CT scan, we draw blood and test it for hormones and specific lipid markers. Patients are then randomly assigned to one of two groups. Members of one group will receive a statin-based cholesterol-lowering medication for three months, while members of the other group will receive a placebo. Then we will repeat the CT scan to see how the amount and structure of the plaque have changed,” explained Dr. Barnabás Baksa.
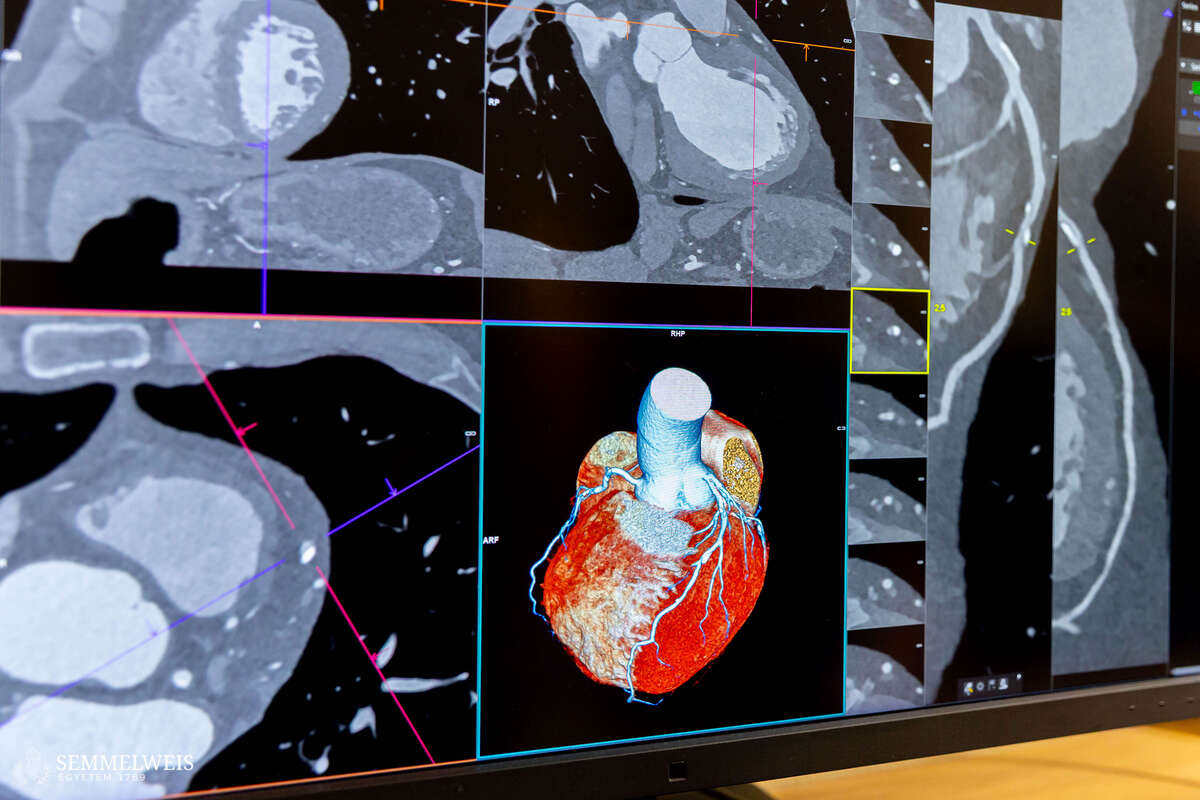
According to Dr. Réka Sebestyén-Dósa, in addition to receiving an extremely detailed picture of the condition of their heart through a photon-counting CT scan, patients also undergo a so-called plaque analysis – a procedure that is unique in Europe and available only as an expensive service in the United States. “This allows us to determine the composition of the plaques not only visually, but also at the level of their structural elements, with cubic-millimeter precision,” the coordinator pointed out.
“We are currently enrolling a total of 140 patients in the clinical trial that began in June 2025. Participation does not require any additional procedures or pose risks for these patients, as the treatment regimen essentially follows what their specialist would have recommended anyway. The treatment is supplemented only by initial and follow-up cardiac CT scans. For half of the participants, conventional statin therapy will begin with a three-month delay. After three months, all patients will receive the statin dose recommended by the cardiologist. What makes our study different is that a unique plaque analysis is performed for every participant. As mentioned earlier, a so-called flow simulation is performed for every single patient enrolling in the clinical trial, which allows us to exclude patients with severe stenosis who require cardiac catheterization and coronary artery dilation; therefore, participation in the clinical trial will pose no risk even to those who receive a placebo during the first three months,” added Dr. Lili Száraz.
The clinical trial also has an important preventive value. It targets people in a very active phase of life who may not be paying enough attention to their health – men aged 40 and older and women aged 45 and older. In their case, cardiovascular problems detected early can prevent serious, even life-threatening heart attacks later. By taking timely precautions and adopting a healthy lifestyle, they can ensure they enjoy a healthy, active old age. – Dr. Lili Száraz
Éva Fekete-Obreczány
Translation: Judit Dőtsch
Photos by Bálint Barta, Attila Kovács – Semmelweis University