Leader: Krenács Tibor, PhD, DSc
The Laboratory is primarily focusing on investigating and validating biomarkers of tumor progression, prognosis and therapy prediciton, and revealing the mechanism of action and otimizing of targetted tumor damage induced by modulated 13,56 MHz radiofrequency generated elctrohyperthermia (mEHT). We utilize in situ methods of molecular morphology (imunohistochemistry, in situ hybridization), high throughput tissue microarray (TMA) method, and automated image analysis supported digital microscopy. When required, our observations are supported by in vitro molecular methods and protein expression techniques, as well as functional tests carried our in cell cultures and animal models
Main projects:
1) Modulated electrohyperthermia (mEHT): Investigating the molecular mechanism of action of mEHT induced targetted tumor cell death and otimizing the effect of combined mEHT+ targetted molecular therapy
Background: Elevated glycolysis and ion concentration in malignant tumors (Warburg effect) result in the selective accumulation of mEHT induced electric field and concommittant heat (<42oC) in tumors leading to their targeted damage.
Model, study material: – In vivo: – xenografts (HT29) and allografts (C26) of colorectal carcinoma cell lines grown in mice – allografts of breast cancer cell lines (tripple negative, or HER2 positive) in mice treated with combined mEHT + immuncheckpoint-ihibitory (Ipilimumab, Nivolumab) or Herceptin therapy – In vitro: – confluent tumor cell lines of mouse and human origine (colorectal, breast and lung cancer) Study focus: – mEHT induced, cell-type dependent programmed cell death response pathways – damage associated molecular pattern (DAMP) signals and immunogenic factors released – the induced anti-tumor immune response – systhemic, distant (abscopal) effect of combined therapy protocols – the significance of glucose metabolism of treated tumors on the efficiency of mEHT therapy – tumorselectivity of mEHT treatment
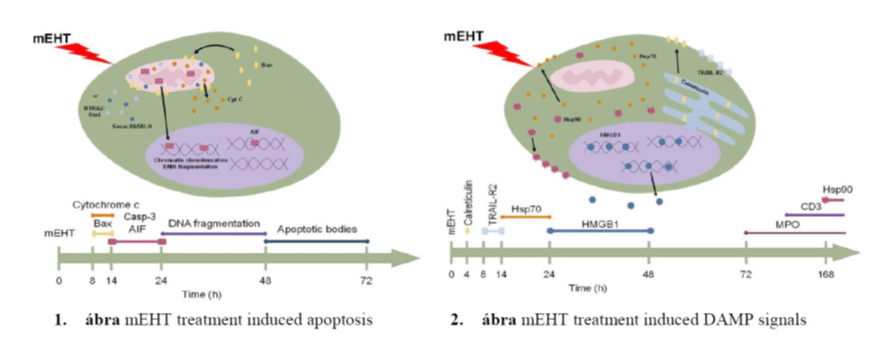
Main results: In colorectal cancer xeno- and allograft models mEHT treatment can result in either caspase-dependent, or -independent (apoptosis inducing factor – AIF mediated) programmed cell death depending on the differential genetic and epigenetic make-up of cancer cells (Figure 1). The treatment also caused the translocation and release of stress associated DAMP proteins (calreticulin, HSP70 and HMGB1) (Figure 2) accompanied by the significant infiltration of the treated tumors by CD8+ cytotoxic T-cells and S100+ antigen presenting cells.
Relevant papers:
1. Andocs G, Meggyeshazi N, Balogh L, Spisak S, Maros ME, Balla P, Kiszner G, Teleki I, Kovago C, Krenacs T. Upregulation of heat shock proteins and the promotion of damage-associated molecular pattern signals in a colorectal cancer model by modulated electrohyperthermia. Cell Stress Chaperones. 2015, 20:37-46.
2. Meggyeshazi N, Andocs G, Balogh L, Balla P, Kiszner G, Teleki I, Jeney A, Krenacs T. DNA fragmentation and caspase-independent programmed cell death by modulated electrohyperthermia. Strahlenther Onkol. 2014, 190:815-822.
2) Investigating biomarkers of tumor progression, prognosis and therapy prediciton
A) In situ study of cell cycle regulation in tumors
Background: Deregulation of cell growth pathways and cell cycle regulation both lead to the autonomous replication of malignant tumors. Elevated cell cycle progression indicates agressive tumor behavior, thus it is linked to poor outcome and may also predict the efficiency of chemotherapy.
Model, study material:
– human melanocytic tumors: nevus, dysplastic nevus, thin melanoma, thick melanoma and metastastativ
melanoma
– humán cultured melanoma (A2058, WM35, HT168, ill. HT199) cell lines and melanocytes
– tissue samples of primary and recurrent giant cell tumors of bone (GCTB) from a cooperation
(EuroBonet, EU FP6) with clinical follow up data, and cultured primary neoplastic GCBT stromal cells.
– osteosarcoma tissue samples with clinical follow-up including treatment data
– breast cancers and head-and-neck cancers with clinical follow-up data from cooperations
Study focus:
Correlations between tumor cell cycle fractions and
– tumor progression as well as
– tumor prognosis (disease free and overall survival)
Main results:
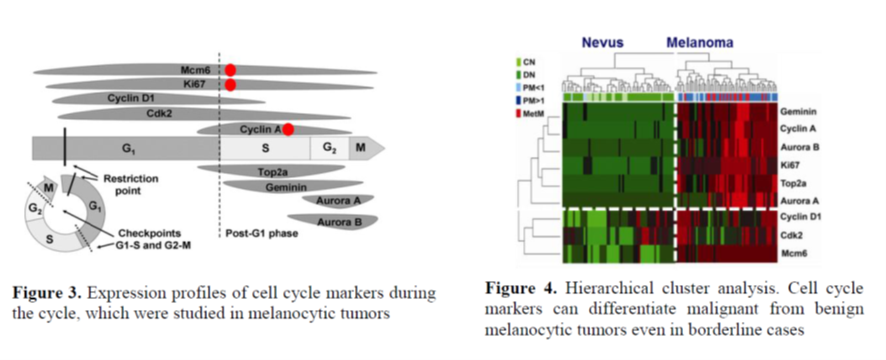
Selected cell cyle markers (Figure 3) can differentiate between malignant and bening melanocytic tumors (Figure 4). In nevi and dysplastic nevi all tumor cells entering the cycle are arrested in the G1-phase. Significant elevation of general cell cycle marker (Mcm6 és Ki67) positive tumor cell fractions and the occurance of the post-G1 phase (cyclin A+) tumor cells differentiate thin melanomas from dysplastic nevi. In thick melanomas cell cycle progression is significantly accelerated compared to thin melanomas. The proliferative phenotype predisposing to metastatic spreading can already be detected in thick melanomas. Expression of Ki-67, MCM2, cyclin A, and PHH3 can predict complete pathological remission in breast cancers. Our study of the link between GCTB progression and cell cycle regulation is under publication; and our work on studying cell cycle regulation in osteosarcoma is still running.
Relevant papers:
1) Kiszner G, Wichmann B, Nemeth IB, Varga E, Meggyeshazi N, Teleki I, Balla P, Maros ME, Penksza K, Krenacs T. Cell cycle analysis can differentiate thin melanomas from dysplastic nevi and reveals accelerated replication in thick melanomas. Virchows Arch. 2014, 464:603-612.
2) Tőkés T, Tőkés AM, Szentmártoni G, Kiszner G, Madaras L, Kulka J, Krenács T, Dank M. Expression of cell cycle markers is predictive of the response to primary systemic therapy of locally advanced breast cancer. Virchows Arch. 2016, 468:675-686.
2/B) Collagen XVII (COL17A1) expression in malignant tumors
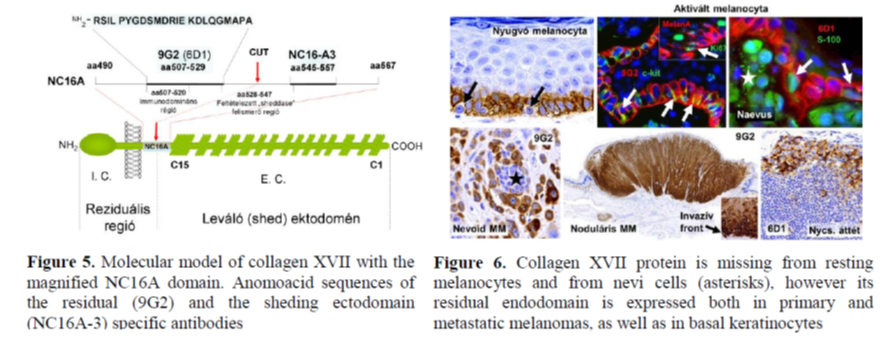
Background: Collagen XVII is a hemidesmosomal protein, which anchores undifferentiated basal keratinocytes to the basement membrane. Normally, the expression of the protein is ceased upon keratinocyte differentiation. However, upon malignant transformation this negative feedback mechanism is deregulated leading to collagen XVII neo-expression as an early indication of malignization. We, for the first time, also observed an upregulated collagen XVII expression in melanomas.
Model, study material:
– human melanocytic tumors: nevus, dysplastic nevus, thin melanoma, thick melanoma and metastatic
melanoma
– humán cultured melanoma (A2058, WM35, HT168, ill. HT199) cell lines and melanocytes
– clinically followed-up breast cancers in cooperation (University of Qeensland, Australia)
Study focus:
– the correlation between collagen XVII expression and tumor progression using ecto- and endodomén
specific antibodies
– detecting mRNA and protein levels in melanocyte cell lines
– the effect of specific antibody treatment of melanoma cell lines
– detection of protein levels at primary and metastatic sites of melanoma (HT199) xenografts
– the link between protein expression and gene romoter hypermethylation during breast cancer
progression
Main results:
The 60 kDa residual domain of collagen XVII protein (Figure 5) can not be detected in resting melanocytes, but it is expressed in malignant melanomas particularly at their invasive fronts (Figure 6). In culture, melanoma growth and migration can be inhibited by binding specific antibody (9G2) to the extracellular region of the residual NC16A endodomain. While the expression of collagen XVII protein is upregulated in epidermal and cervical squamous cell carcinomas, it is downregulated in breast cancer compared to nomal breast epithelium due to gene promoter hypermethylation resulting in an increased invasive potential.
Relevant papers:
1) Thangavelu PU, Krenacs T, Dray E, Duijf PH. In epithelial cancers, aberrant COL17A1 promoter methylation predicts its misexpression and increased invasion. Clin Epigenetics. 2016 Nov 18;8:120.
2) Krenacs T, Kiszner G, Stelkovics E, Balla P, Teleki I, Nemeth I, Varga E, Korom I, Barbai T, Plotar V, Timar J, Raso E. Collagen XVII is expressed in malignant but not in benign melanocytic tumors and it can mediate antibody induced melanoma apoptosis. Histochem Cell Biol. 2012, 138:653-667.
3) Stelkovics E, Korom I, Marczinovits I, Molnar J, Rasky K, Raso E, Ficsor L, Molnar B, Kopper L, Krenacs T. Collagen XVII/BP180 protein expression in squamous cell carcinoma of the skin detected with novel monoclonal antibodies in archived tissues using tissue microarrays and digital microscopy. Appl Immunohistochem Mol Morphol. 2008, 16:433-441.
2/C Connexin expression and chanel functions during tumor progression
Background: The tetraspan connexin proteins oligomerized into hexameric channels can mediate direct cell-cell communication between adjacent cells by excluding the extracellular space. These communication channels can transport ions and <1,8 kDa molecules including metabolits, morphogens and secondary messengers. Connexin channels play important roles in morphogenesis, tissue regeneration and in synchronizing adaptive cellular responses. Connexin expression and functions are reduced during tumor development and progression, however, re-expression is also observed during tumor invasion and metastasis.
Model, study material:
– tissue samples of primary and recurrent giant cell tumors of bone (GCTB) from a cooperation
(EuroBonet, EU FP6) with clinical follow-up data, and cultured primary neoplastic GCBT stromal cells
– low density cell fractions isolated from reactive human tonsils cultured for forming ex vivo lymphoid
follicles; and follicular lymphomas
– In silico database analysis of connexin mRNA expression tested with Affimetrix and Illumina platforms
in breast cancer
– breast cancer series with clinical follow-up data from national (SE, 2nd Department of Pathology) and
international (Zürich-i Egyetem) cooperations
Study focus:
– connexin43 (Cx43) expression and channel functions in GCTB tumor progression
– inhibition of Cx43 expression and channel functions using Gap27 connexin mimetic peptide in culture
during ex vivo lymphoid follicle formation
– Cx43 expression and follicular lymphoma (FL) progression
– connexin isotype expression and breast cancer progression and prognosis
– connexin expression during primary systemic (neoadjuvant) chemotherapy and utilyzing results in
predicting treatment efficiency
Main results:
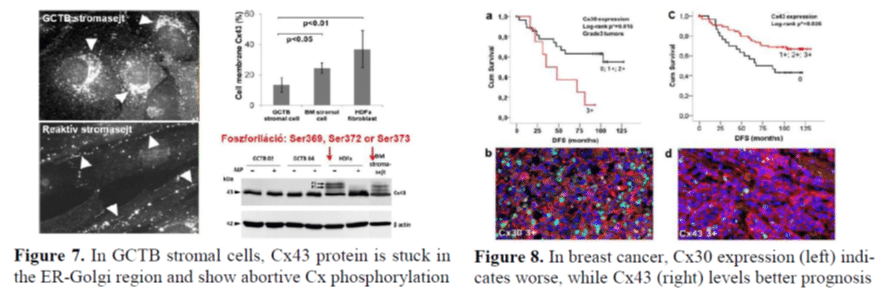
Deregulation of Cx43 cannel phosphorylation and funtions (Figure 7) in GCTB neoplastic stromal cells and the reduced osteoclastogenic control contribute to clinical tumor progression and worse prognosis. Inhibition of Cx43 channel docking and functions using connexin mimetic peptide results in the formation of abortive ex vivo lymphoid follicles and the devastation of follicular dendritic cells and B-cells, however Cx43 expression does not show a significant link with FL progression. In breast cancer, connexin isotypes show cancer type/stage related selective expression, and may function either as tumor suppressors (e.g Cx43 and Cx46), or tumor promoters (Cx30) (Figure 8). Therefore, detection of connexin isotypes can serve either as independent positive, or negative prognostic factors.
Relevant papers:
1. Balla P, Maros ME, Barna G, Antal I, Papp G, Sapi Z, Athanasou NA, Benassi MS, Picci P, Krenacs T. Prognostic impact of reduced connexin43 expression and gap junction coupling of neoplastic stromal cells in giant cell tumor of bone. PLoS One. 2015 May 1;10(5):e0125316.
2. Rajnai H, Teleki I, Kiszner G, Meggyesházi N, Balla P, Vancsik T, Muzes G, Csomor J, Matolcsy A, Krenacs T. Connexin 43 communication channels in follicular dendritic cell development and in follicular lymphomas. J Immunol Res. 2015;2015:528098.
3. Teleki I, Szasz AM, Maros ME, Gyorffy B, Kulka J, Meggyeshazi N, Kiszner G, Balla P, Samu A, Krenacs T. Correlations of differentially expressed gap junction connexins Cx26, Cx30, Cx32, Cx43 and Cx46 with breast cancer progression and prognosis. PLoS One. 2014 Nov 10;9(11):e112541.
4. Teleki I, Krenacs T, Szasz MA, Kulka J, Wichmann B, Leo C, Papassotiropoulos B, Riemenschnitter C, Moch H, Varga Z. The potential prognostic value of connexin 26 and 46 expression in neoadjuvant-treated breast cancer. BMC Cancer. 2013 Feb 2;13:50.