Brain and Cognition 91 (2014) 62–70
DOI: 10.1016/j.bandc.2014.08.004
Péter Simora,b János Körmendic, Klára Horváthd, Ferenc Gombose, Péter P. Ujmac, Róbert Bódizsc,e
a Department of Cognitive Sciences, Budapest University of Technology and Economics, H-1111, Egry József u. 1, T. épület/V, Budapest, Hungary
b Nyírő Gyula Hospital, National Institute of Psychiatry and Addictions, Budapest, Hungary
c Institute of Behavioural Sciences, Semmelweis University, H-1089, Nagyvárad tér 4, Budapest, Hungary
d Department of Experimental Psychology, University of Oxford, South Parks Road, OX1 3UD Oxford, UK
e Department of General Psychology, Pázmány Péter Catholic University, 2087, Egyetem u. 1, Piliscsaba, Hungary
Corresponding author at: Department of Cognitive Sciences, Budapest University of Technology and Economics, H-1111, Egry József u. 1, T. épület/V, Budapest, Hungary. Fax: +36 1 463 1072. E-mail addresses: psimor@cogsci.bme.hu, petersimor@gmail.com (P. Simor).
Abbreviations:
AI, atonia index; ANCOVA, analysis of covariance; CAP, cyclic alternating pattern; CTL, control; DQQ, Dream Quality Questionnaire; ECG, electrocardiogram; EEG, electroencephalography; HF, high frequency measure of heart rate variability; HRV, heart rate variability; LF, low frequency measure of heart rate variability; NM, nightmare; NN, normal sinus to normal sinus; PTSD, post traumatic stress disorder; REM, non-rapid eye movement sleep; REM, rapid eye movement sleep; RMSSD, square root of the mean of the squares of differences between adjacent normal sinus to normal sinus intervals; VDAS-H, the Hungarian version of the Van Dream Anxiety Scale.
Abstract
Abnormal arousal processes, sympathetic influences, as well as wake-like alpha activity during sleep were reported as pathophysiological features of Nightmare Disorder. We hypothesized that in Nightmare Disorder, wake-like cortical activity and peripheral measures linked to arousals would be triggered by physiological processes related to the initiation of REM periods. Therefore, we examined electroencephalographic (EEG), motor and autonomous (cardiac) activity in a group of nightmare (NM) and healthy control (CTL) subjects during sleep-state-transitions while controlling for the confounding effects of trait anxiety. Based on the second-nights’ polysomnographic recordings of 19 Nightmare Disordered (NM) and 21 control (CTL) subjects, we examined the absolute power spectra focusing on the alpha range, measures of heart rate variability (HRV) and motor (muscle tone) activity during pre-REM and post-REM periods, separately. According to our results, the NM group exhibited increased alpha power during pre-REM, but not in post-REM, or stable, non-transitory periods. While CTL subjects showed increased HRV during pre-REM periods in contrast to post-REM ones, NM subjects did not exhibit such sleep state-specific differences in HRV, but showed more stable values across the examined sleep stages and less overall variability reflecting generally attenuated parasympathetic activity during sleep-state-transitions and during stable, non-transitory NREM states. These differences were not mediated by waking levels of trait anxiety. Moreover, in both groups, significant differences emerged regarding cortical and motor (muscle tone) activity between pre-REM and post-REM conditions, reflecting the heterogeneity of NREM sleep. Our findings indicate that NM subjects’ sleep is compromised during NREM–REM transitions, but relatively stabilized after REM periods. The coexistence of sleep-like and wake-like cortical activity in NM subjects seems to be triggered by REM/WAKE promoting neural activity. We propose that increased arousalrelated phenomena in NREM–REM transitions might reflect altered emotional processing in NM subjects.
Keywords: Sleep, Nightmare, Dreaming, EEG, Alpha power, Heart rate variability
- Introduction
Although at the behavioral level sleep is apparently a global, whole-brain phenomenon that is governed by central mechanisms (Saper, Chou, & Scammell, 2001), neural oscillations during sleep often indicate the coexistence of markedly different EEG patterns, especially during the transitions between different states of vigilance (Bódizs, Simor, Csóka, Bérdi, & Kopp, 2008; Bódizs, Sverteczki, & Mészáros, 2008; De Gennaro, Ferrara, Curcio, & Cristiani, 2001; Nobili et al., 2011). The coexistence of different sleep-like and wake-like electroencephalographic (EEG) oscillations was shown to be related to lucid dream experiences (Tyson, Ogilvie, & Hunt, 1984; Voss, Holzmann, Tuin, & Hobson, 2009), arousal parasomnias (Terzaghi et al., 2009), occasional sleep paralysis (Mahowald, Cramer Bornemann, & Schenck, 2011), and sleep paralysis within the context of narcolepsy (Terzaghi, Ratti, Manni, & Manni, 2012), or REM Behavior Disorder (Manni, Terzaghi, & Glorioso, 2009).
More recently, a relatively less emphasized sleep disorder, Nightmare Disorder was also characterized by the combination of wake-like and sleep-like EEG features during REM sleep (Simor, Horváth, Ujma, Gombos, & Bódizs, 2013). Subjects with Nightmare Disorder report at least one emotionally negative dream on a weekly basis; which experiences may result in abrupt awakenings from (usually) late-night sleep (ICSD-II; American Sleep Disorders Association, 2005). Higher arousability, and lower awakening thresholds in Nightmare Disorder seem to be related to the qualitative aspects of dream experiences. For instance, nightmares that end with abrupt awakenings in contrast to dysphoric dreams without awakenings were rated by dreamers as emotionally more intense, containing a higher rate of physical aggression and more negative scenario (e.g. failures and unfortunate endings) (Robert & Zadra, 2014; Zadra, Pilon, & Donderi, 2006). Frequent nightmare complaints are highly prevalent, affecting approximately four percent of the adult population (Spoormaker, Schredl, & van den Bout, 2006). The clinical relevance of nightmares should not be underestimated given their high incidence in psychiatric populations (Swart, van Schagen, Lancee, & van den Bout, 2013), co-morbidity with insomniac symptoms (Schredl, 2009; Schredl, Schafer, Weber, & Heuser, 1998) and their association with waking affective (Levin, Fireman, Spendlove, & Pope, 2011; Sjöström, Hetta, & Waern, 2009; Van Liempt, van Zuiden, Westenberg, Super, & Vermetten, 2013) as well as cognitive (Simor, Pajkossy, Horváth, & Bódizs, 2012) dysfunctions.
Polysomnographic studies suggest that subjects with frequent nightmares in comparison with controls are characterized by disturbed sleep patterns, comprising frequent nocturnal awakenings (Simor, Horváth, Gombos, Takács, & Bódizs, 2012) and enhanced microarousals (Simor, Bódizs, Horváth, & Ferri, 2013) during NREM sleep, increased sympathetic (cardiac) activation (Nielsen et al., 2010) and heightened wake-like alpha power during REM sleep (Simor, Horváth, et al., 2013), as well as elevated indices of periodic leg movements (Germain & Nielsen, 2003) during both sleep stages. However, the underlying mechanism of disrupted sleep in relation to frequent nightmares is far from being fully elucidated.
Sleep stage transitions are specifically ‘‘sensitive’’ periods for the presence of microarousals, muscle activity and EEG desynchronization (Pilcher & Schulz, 1987). These transitory periods clearly show the heterogeneity of sleep as well as the constraints of defining discrete sleep stages. The ambiguity of sleep stages is particularly prevalent in children’s (Grigg-Damberger et al., 2007) and in clinical patient’s sleep recordings (Mahowald et al., 2011). The coexistence of different sleep and awake states is relatively frequent during the so-called ascending phases of sleep, comprising the transition from NREM to REM sleep (Halász & Bódizs, 2013; Parrino, Ferri, Bruni, & Terzano, 2012). During these periods, the cortex is prone to exhibit phasic arousals involving wake-like EEG (McKinney, Dang-Vu, Buxton, Solet, & Ellenbogen, 2011), increased motor activity (Nobili et al., 2011) and autonomic fluctuations (Halász & Bódizs, 2013). During the so-called ascending phases (pre-REM periods) spontaneous or elicited micro-arousals are more frequent and involve the desynchronization of EEG as well as associated autonomic changes (Parrino et al., 2012). In contrast, during the so-called descending slopes (post-REM periods) phasic activations are rare, and environmental stimulation is more likely to induce a K-complex, a sleep-like (antiarousal) response (Halász & Bódizs, 2013). Moreover, EEG spectral power timecourses over the ultradian sleep cycle (Merica & Fortune, 1997, 2004) indicate that the ascending, in comparison to the descending phases, are characterized by reduced sleep promotion, increased environmental awareness, and presumably the intensification of mental (dream) experiences (Conduit, Bruck, & Coleman, 1997; De Gennaro et al., 2010).
We assume that nightmare sufferers’ sleep is particularly vulnerable during the ascending, transitory pre-REM periods, and will show signs of covert wake-like activity, in contrast to the descending, post-REM periods, when due to the reduction of REM sleep pressure, and the reinstatement of sleep-promoting influences, the brain gradually advances into deeper and less fragile sleep stages. In order to examine the cortical correlates of wake-like neural activity we focused on the EEG alpha band power that was shown to reveal instantaneous responsiveness to environmental stimulation as well as spontaneous fluctuations in relation to sleep fragility (McKinney et al., 2011). Since alpha band power seems to reflect covert wake-like activity and increased environmental awareness during sleep (McKinney et al., 2011; Terzaghi et al., 2009, 2012), we hypothesized that nightmare (NM) in comparison to control subjects (CTL) would exhibit increased EEG alpha power, especially during the pre-REM periods. In addition, we examined two ‘‘peripheral’’ measures related to arousals and sleep disruption: heart rate variability (HRV) reflecting parasympathetic control on cardiac regulation (Ferini-Strambi et al., 2000) and muscle atonia (Ferri et al., 2010) indexing motor (in)activity during sleep.
The analysis of HRV is a widely used method for evaluating autonomic cardiovascular regulation (Heart Rate Variability, 1996) characterizing different states of vigilance (Elsenbruch, Harnish, & Orr, 1999) and showing alterations in different sleep disorders, such as insomnia (Spiegelhalder et al., 2011; Vgontzas, Fernandez-Mendoza, Liao, & Bixler, 2013), PTSD (Chang et al., 2013) and Nightmare Disorder (Nielsen et al., 2010). Frequency and time domain measures of HRV based on the variability of interbeat intervals are reliable indices of parasympathetic activity related to the vagal tone (Heart Rate Variability, 1996; Reyes del Paso, Langewitz, Mulder, van Roon, & Duschek, 2013). Since wake-like oscillations and arousals during sleep are related to reduced parasympathetic (vagal) tone (Ferini-Strambi et al., 2000), we expected that during pre-REM, in contrast to post-REM periods, NM subjects would exhibit reduced parasympathetic control in comparison with the control group.
The automatic quantification of muscle atonia, based on the submentalis (chin) muscle EMG recording proved to be an efficient tool to describe abnormal motor activity during REM sleep (Ferri, Bruni, Fulda, Zucconi, & Plazzi, 2012). We hypothesized that NM in comparison to CTL subjects would show signs of abnormal motor activity (reduced muscle atonia) in the pre-REM, but not in the post-REM periods.
Given that waking levels of anxiety were reported to be moderately associated with nightmare frequency (Schredl, 2003), but may also have an effect on EEG spectral power (Mizuki, Kajimura, Nishikori, Imaizumi, & Yamada, 1984; Suetsugi, Mizuki, Ushijima, Yamada, & Imaizumi, 1998), HRV (Dishman et al., 2000; Gorman & Sloan, 2000; Kemp, Quintana, Felmingham, Matthews, & Jelinek, 2012) and muscle activity (Willmann, Langlet, Hainaut, & Bolmont, 2012), we aimed to control for the confounding effects of trait anxiety. Although nightmare frequency and increased anxiety often appear as co-morbid symptoms, research indicates that these variables are indeed independent from each other, and should be treated as separate factors, especially in case of Nightmare Disorder (Coolidge, Segal, Coolidge, Spinath, & Gottschling, 2010; Lancee, Spoormaker, & Van den Bout, 2010; Simor, Horváth, et al., 2012; Spoormaker et al., 2006; Wood & Bootzin, 1990). Therefore, by controlling for the influence of trait anxiety, we could examine the primary relationship between disturbed dreaming and sleep physiology (EEG, HRV and muscle tone), regardless of the effects of waking anxiety.
2. Materials and methods
2.1. Participants
Participants (all native Hungarians) were selected from a large pool of undergraduate students from the Budapest University of Technology and Economics and Semmelweis University. None of the subjects reported prior neurological, psychiatric or sleep disorders or prior history of any chronic disease. 21 NM and 23 CTL subjects took part in polysomnographic examination; however, two NM subjects left the experiment after the first (baseline) night and two of the CTLs’ recordings were considered too noisy for spectral analyses. Therefore, 19 NM (10 males; Mage = 20.94 ± 1.58) and 21 CTL subjects (11 male; Mage = 21.57 ± 1.47) were included in the present study. There was no significant age difference between the two groups (t(38) = 1.3; p = 0.2). The study protocol was approved by the Ethical Committee of the Semmelweis University. The subjects received monetary compensation (approximately 20 EUROS) for their participation in the sleep laboratory investigations. Written informed consent was obtained.
2.1.1. Psychometric instruments for the selection of participants
NM and CTL subjects were enrolled after a stringent selection procedure described previously in detail (Simor, Horváth, et al., 2012). In brief, subjects were selected based on their scores on three different dreaming-related questionnaires: the Dream Quality Questionnaire (DQQ) (Bódizs, Simor, et al., 2008; Bódizs, Sverteczki, et al., 2008), the Hungarian version of the Van Dream Anxiety Scale (VDAS-H) (Simor et al., 2009) and a 7-point Likert scale assessing the frequency of nightmares. Subjects reporting one or more nightmares per week were assigned to the NM group, while individuals having less than two nightmares during the last year were assigned as CTL subjects. Those subjects who reported the onset of negative dream experiences in relation to a traumatic event or indicated that the content of their dreams were related to a prior trauma (such as physical attack, accident and sudden death of a close relative) were excluded from the study.
NM subjects scored higher on the Negative Dream Affect Scale of the DQQ (MNM = 8.12; SDNM = 1.91 vs. MCTL = 4.03; SDCTL = 1.77; t(38) = 7.25; p < 0.0001) and on the VDAS-H (MNM = 20.58; SDNM = 7.5 vs. MCTL: 0.23 ± SDCTL = .62; t(18.22) = 11.73; p < 0.0001; equal variances not assumed), indicating at least moderately severe dream disturbances (Bódizs, Simor, et al., 2008; Bódizs, Sverteczki, et al., 2008; Simor et al., 2009). To statistically control the confounding effects of waking anxiety on EEG, EMG and ECG measures, subjects were asked to complete the Hungarian version of the Spielberger State-Trait Anxiety Inventory (only the trait measure STAI-T) (Sipos, Sipos, & Spielberger, 1994). NM in comparison with CTL subjects showed significantly higher scores on trait levels of anxiety (MNM = 50.42 ± 8.15 vs. MCTL = 33.28 ± 8.54; t(38) = 6.85; p < 0.001), therefore, in order to partial out the confounding effects of anxiety on our dependent measures, in the subsequent analyses we controlled the effects of STAI-T scores on our variables of interest.
2.2. Procedure
Polysomnographic recordings were performed for two consecutive nights. (The first night served as the adaptation night.) Subjects were not allowed to drink alcohol or take drugs (except contraceptives) on the day and the previous day of the examination. They were asked to avoid napping and consuming caffeine in the afternoon of the sleep recordings. The timing of lights off was between 11.00 PM and 1.00 AM depending on each participant’s preferred bedtime. Morning awakenings were scheduled after 9 h of undisturbed sleep unless participants woke up earlier spontaneously.
2.3. Polysomnography
On both nights, subjects were fitted with 19 EEG electrodes (Fp1, Fp2, F3, F4, Fz, F7, F8, C3, C4, Cz, P3, P4, Pz, T3, T4, T5, T6, O1, O2) according to the 10–20 electrode placement system (Jasper, 1958) as well as with two EOG electrodes (bipolar channel) monitoring vertical and horizontal eye-movements; EMG electrodes (bipolar channels) for the chin and for the anterior tibialis muscles, two ECG electrodes according to standard lead I; in addition to the thoracic and abdominal respiration sensors. Goldcoated Ag/AgCl EEG cup electrodes were fixed with EC2 Grass Electrode Cream (Grass Technologies, USA) and referred to the mathematically-linked mastoids. Impedances were kept below 8 kX. Signals were collected, prefiltered (0.33–1500 Hz, 40 dB/decade anti-aliasing hardware input filter), amplified and digitized with 4096 Hz/channel sampling rate (synchronous) with 12 bit resolution by using the 32 channel EEG/polysystem (Brain-Quick BQ 132S, Micromed, Italy). A further 40 dB/decade anti-aliasing digital filter was applied by digital signal processing which low-pass filtered the data at 450 Hz. Finally, the digitized and filtered EEG was undersampled at 1024 Hz.
2.4. Data analysis
Sleep stages were scored according to standardized criteria (Iber, Ancoli-Israel, Chesson, & Quan, 2007) by two experienced sleep researchers who were blind to the group membership of the participants. Pre-REM and post-REM periods were selected from each subjects’ recording for further analyses. Pre-REM periods were defined as 10-min intervals of NREM sleep (including Stage 2 and SWS) directly before the onset of REM periods. Accordingly, post-REM periods included similar 10-min long NREM epochs following the end of REM periods. Overlapping (50%), artifact-free four-second-epochs of all EEG derivations were Hanning-tapered and Fourier transformed by using the FFT (Fast Fourier Transformation) algorithm in order to calculate the average power spectral density (lV2/0.25 Hz) for pre-REM and post-REM periods, separately. Absolute spectral power values were obtained for each frequency bin (width: 0.25 Hz) at each recording site within the range of 1–48 Hz. The alpha band power was computed as the sum of the bin-wise power values between 8 and 13 Hz, averaged across all derivations. Bin-wise absolute spectral power as well as alpha band power values were log-transformed by using a 10 base logarithm in order to normalize their distribution before performing statistical analyses.
Analysis of HRV was performed by the HRV toolkit for MATLAB developed by Kaplan and Staffin (Software for Heart Rate Variability, http://www.macalester.edu/~kaplan/hrv/doc/). The same 10min-long segments of pre-and post-REM periods were selected from the ECG signal. R waves were automatically detected and visually screened in order to correct missed detections, as well as to exclude technical artifacts and arrhythmias. Because of these artifacts the length of these 10-min-long segments was shortened. The minimum length of the selected and analyzed segments was set to 5 min. If the segment without artifacts was shorter than this minimum value, it was excluded from the analyses. (Each subject had at least three separate pre-and post-REM segments to be analyzed.) R–R variability of normal sinus to normal sinus (NN) interbeat intervals was analyzed. We computed the low (LF: 0.04– 0.15 Hz) and the high frequency (HF: 0.15–0.4 Hz) component of HRV, as well as the LF/HF ratio, and the time domain measure RMSSD (square root of the mean of the squares of differences between adjacent NN intervals) for each segment, in each subject.
Finally, we averaged these values across the analyzed pre-and post-REM segments, separately.
In order to measure muscle activity, we calculated the chin EMG atonia index (Ferri et al., 2012). In brief, band-pass (butterworth zero phase forward and reverse digital filter; 10–100 Hz) filtered submentalis muscle EEG signals from the pre-and post-REM periods were selected, and after the removal of technical artifacts, signals were rectified and subdivided into one-second mini epochs. The average amplitude was obtained for each mini epoch, and subsequently a noise correction method was applied by subtracting the minimal value from each mini epoch found in a moving window including ±30 s surrounding it (for the detailed description of the procedure see Ferri et al. (2010)). The EMG atonia index (AI) was computed for all pre-and post-REM periods. The AI shows the ratio of EMG mini epochs with amplitude 61 lV to the total number of epochs excluding the mini epochs with 1 lV < amplitude 6 2 lV. Finally, the AI was averaged across the selected preREM and post-REM periods, separately. This way, we obtained a pre-REM and post-REM AI score for each subject. Lower values indicate reduced muscle atonia and increased motor activity. The data of one CTL subject was excluded because of too many technical, noise-related artifacts in the chin EMG recording.
Although the main purpose of the present study was to compare transient sleep periods in NMs and CTLs, in order to examine the specificity of our results, we computed EEG alpha power, HRV and muscle atonia index during non-transitory NREM periods as well. Non-transitory periods were defined as NREM segments free of any stage shifts and beginning 20 min after the end of REM sleep and terminating before 20 min of the next REM sleep period. Nontransitory NREM segments from the first 3 sleep cycles were randomly selected for EEG, EMG and ECG data. The length of the periods was same as the pre-and post REM segments.
Groups did not significantly differ regarding sleep latency (t(38) = 1.6; ns), REM latency (t(38) = 0.69; ns) or sleep cycle duration (t(38) = 0.72; ns), and the time of the night of the selected segments was comparable (t(38) = 0.89; ns) between the two groups.
2.5. Statistics
Statistical analyses were carried out with the Statistical Package for the Social Sciences version 19.0 (IBM) and MATLAB (version 7.10.0.499, R2010a, The MathWorks, Inc., Natick, MA). We performed separate Repeated Measure Analyses of Covariance (ANCOVA) in order to examine the main effects of Group (NM vs. CTL), Condition (pre-REM vs. post-REM) as well as their interaction (Group Condition) on the (six) dependent variables of interest. In order to control for the confounding effects of anxiety on EEG, EMG and ECG measures, the STAI-T scores were entered in all cases as covariates in the statistical models. This way, EEG alpha power, EMG Atonia Index (AI), and the different HRV measures (LF, HF, LF/HF, RMSSD) were entered in six separate Repeated Measures ANCOVA models. The issue of multiple comparisons was addressed by False Discovery Rate (FDR) correction (Benjamini, Drai, Elmer, Kafkafi, & Golani, 2001). The FDR method takes into account the correlations among the examined variables, and can handle type 1 and type 2 errors when p-values come from positively correlated variables. Therefore, in our case the FDR procedure is more adequate in comparison to other widely used statistical corrections (eg. Bonferroni) that assume the individual tests to be independent from each other which is not the case for EEG, HRV and EMG measures.
In order to examine the specificity of EEG alpha power reflecting Group differences between pre-and post-REM periods, we also performed bin-wise (0.25 Hz) spectral power analyses. We entered absolute spectral power values of each electrode and frequency bin (1–48 Hz) as dependent, Group membership as fixed, and STAI-T scores as covariate variables in multiple univariate ANCOVA model for pre-and post-REM sleep, separately. In this case, multiple comparisons were corrected by the procedure of descriptive data analysis, delineating the so called Rüger’s areas (Abt, 1988; Duffy et al., 1990). The procedure is designed to handle data with strongly intercorrelated neighboring data points. Rüger’s areas are defined as sets of conventionally significant (p < 0.05) results which are accepted or rejected as significant as a whole, instead of individual results of statistical tests. We took the results of our statistical tests as a two-dimensional matrix (with electrodes being one dimension, and frequency bins the other) and defined Rüger’s areas along the dimension of frequency bins. A Rüger’s area started when there was a significant result on any electrode in that frequency bin, continued into higher frequencies, and ended wherever there were no significant results on any electrode. After defining these areas of significance, a frequency range was considered as significant if at least half of the conventional (p = 0.05) significance level were below p/2 = 0.025 and at least one third of them were below p/ 3 = 0.0167. Single-bin results were not considered as an area. This conservative method addresses the issue of multiple comparisons since the probability that all these requirements will be fulfilled by chance is low.
3. Results
3.1. EEG absolute alpha power
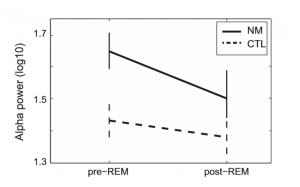
Regarding EEG absolute alpha power values, a significant main effect of Condition (F(1, 37) = 25.1; p < 0.0001) and a significant Group Condition (F(1, 37) = 9.84; p = 0.003) interaction emerged. Pairwise comparisons indicated increased alpha power values during pre-REM in contrast to post-REM periods (Estimated Marginal Mean (EMM)pre-REM: 1.54, Standard Error (SE): 0.3 vs. EMMpost-REM: 1.44, SE: 0.31; p < 0.0001). The main effect of Group showed a trend (F(1, 37) = 3.65; p = 0.06). Post hoc comparisons of Group by Condition, based on univariate ANCOVA showed significantly increased alpha power in NM during pre-REM (EMMNM = 1.65, SE: 0.55 vs. EMMCTL = 1.43, SE: 0.52; F(1, 37) = 6.045; p = 0.019), but a relatively less robust difference during post-REM periods (EMMNM = 1.5, SE: 0.57 vs. EMMCTL = 1.38, SE: 0.53; F(1, 37) = 1.75; not significant (ns). In sum, these data indicate that NM in comparison with CTL subjects are characterized by increased alpha power during pre-REM periods; however, following REM phases, during post-REM periods, alpha power is reduced, approximating control values (see Fig. 1).
To examine the specificity of the alpha band differentiating the two groups in the pre-and post-REM conditions, we carried out bin-wise spectral power analyses for the whole spectrum applying multiple univariate ANCOVA with absolute spectral power values of each frequency bin (1–48 Hz) at each scalp derivation as the dependent variables, Group as the fixed, and STAI-T as the covariate in the statistical models. In the pre-REM condition, the NM group was characterized by significantly higher absolute power values in the 7–12.25 Hz frequency range in almost all recording sites (Fp1, Fp2, F7, F3, Fz, F4, F8, C3, Cz, C4, T4, Pz, P4, T6, O1, O2). These differences remained significant after correction for multiple comparisons. Significant differences were also observed in the 17.75–27.25 Hz (beta) frequency range in pre-REM periods, with the NM group exhibiting higher activity in several derivations (Fp1, Fp2, F7, F3, F4, F8, C3, T4, T5, Pz, P4, T6, O1, O2); however, these differences did not survive the stringent correction for multiple comparisons. In contrast to pre-REM periods, during post-REM periods, no significant differences emerged between the two groups.
Fig. 1. Absolute alpha power values of nightmare and control subjects during preREM and post-REM periods. Estimated Marginal Means (STAI-T = 41.43) of log transformed (log 10) alpha power values as well as standard errors are plotted. CTL – control group; NM – nightmare group.
3.2. HRV measures
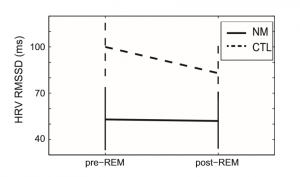
The mean heart rate of all subjects was within normal ranges (range: 46.78–73.86). Regarding the LF component of HRV the effect of Condition was not significant (F(1, 37) = 1.7; ns), but the effect of Group as well as the Group Condition interaction showed a trend (F(1, 37) = 4.35; p = 0.04 and F(1, 37) = 3.87; p = 0.06, respectively). The HF component clearly differentiated the two groups. A significant main effect for Condition F(1, 37) = 6.6; p = 0.01), Group (F(1, 37) = 8.1; p = 0.007) and Group Condition (F(1, 37) = 9.39; p = 0.004) was observed. No significant effects were evident on the LF/HF ratio. The time domain measure of HRV (see Fig. 2), the RMSSD showed a significant main effect for Group F(1, 37) = 6.43; p = 0.015, a significant Group Condition interaction (F(1, 37) = 7.86; p = 0.008), while the main effect of Condition showed a trend (F(1, 37) = 3.54; p = 0.06.
Fig. 2. The time domain measure of HRV in nightmare and control subjects within pre-REM and post-REM periods. The vertical axis shows the Estimated Marginal Means (STAI-T = 41.43) as well as standard errors of the RMSSD HRV scores. CTL – control group; NM – nightmare group; HRV – heart rate variability; RMSSD – square root of the mean of the squares of differences between adjacent normal sinus-to normal sinus intervals.
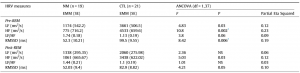
Pairwise group comparisons by multiple univariate ANCOVA are summarized in Table 1. The NM group in comparison to CTL subjects, exhibited reduced power values regarding the LF and HF component as well reduced HRV indexed by the RMSSD score within pre-REM, and reduced HF and RMSSD values within postREM periods. Group differences in HF power and RMSSD score (indexing parasympathetic activity) of the pre-REM condition survived FDR correction for multiple comparisons. It is important to note that CTL subjects showed larger LF, HF and RMSSD scores in the pre-REM condition in contrast to the post-REM condition, while the NM group exhibited more stable values across the preand post-REM periods, especially in measures reflecting parasympathetic activity (HF, RMSSD).
3.3. EMG atonia index (AI)
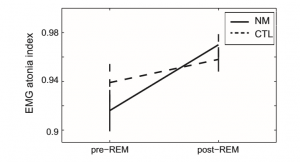
A significant main effect of Condition emerged (F(1, 36) = 9.26; p = 0.003) due to reduced AI scores during pre-REM compared to post-REM sleep (EMMpre-REM: 0.93, SE: 0.009 vs. EMMpost-REM: 0.96, SE: 0.006; p < 0.0001). The main effect of Group was not significant (F(1, 36) = 0.78; ns), and the Group Condition interaction showed a trend (F(1, 36) = 2.77; p = 0.1), as can be observed in Fig. 3. NM in contrast to CTL subjects were characterized by lower muscle atonia in pre-REM (EMMNM = 0.916, SE: 0.017 vs. EMMCTL = 0.939, SE: 0.016), but higher muscle atonia (EMMNM = 0.97, SE: 0.01 vs. EMMCTL = 0.958, SE: 0.01) in postREM periods. Nevertheless, post hoc pairwise comparisons examining independent between-subject effects (applying univariate ANCOVA), showed no significant differences between groups in these conditions.
3.4. Non-transitory NREM periods
Alpha power during non-transitory NREM periods was not significantly different between NMs and CTLs (F(1, 37) = 2.54; ns), (EMMNM = 1.69, SE: 0.07 vs. EMMCTL = 1.53, SE: 0.06). In contrast, HRV measures, including LF, HF and RMSSD differentiated the two groups. NMs showed significantly lower values in LF (F(1, 37) = 8.42; p = 0.006), (EMMNM = 783, SE: 260.8 vs. EMMCTL = 1988, SE: 243.7), HF (F(1, 37) = 9.61; p = 0.004), (EMMNM = 719, SE: 451.2 vs. EMMCTL = 2947, SE: 421.6) and RMSSD (F(1, 37) = 10.91; p = 0.002), (EMMNM = 43.7, SE: 7.05 vs. EMMCTL = 80.8, SE: 6.58) suggesting that NMs exhibited generally reduced parasympathetic cardiac activity during stabile, non-transient NREM periods. The LF/HF ratio did not show significant differences (F(1, 37) = 0.28; p = ns.), (EMMNM = 1.15, SE: 0.19 vs. EMMCTL = 0.99, SE: 0.18). No significant differences emerged regarding muscle atonia (F(1, 37) = 0.77; ns), (EMMNM = 0.98, SE: 0.02 vs. EMMCTL = 0.95, SE: 0.2) during these NREM periods.
Table 1. Comparison of HRV measures during pre-REM and post-REM periods between nightmare and control subjects. Adjusted means, standard errors as well as Group effects of univariate analyses of covariance are shown with STAI-T (41.43) as covariate.
HRV – heart rate variability; LF – low frequency power; HF – high frequency power; RMSSD – square root of the mean of the squares of differences between adjacent normal sinus to normal sinus intervals; NM – nightmare subjects; CTL – control subjects; ANCOVA – Analyses of Covariance; EMM – Estimated Marginal Means; SE – Standard Error; NS – not significant.
* Significant after false discovery rate correction.
Fig. 3. The chin EMG atonia index in nightmare and control subjects within preREM and post-REM periods. The vertical axis shows the Estimated Marginal Means (STAI-T = 41.43) as well as standard errors of the EMG atonia index scores. CTL – control group; NM – nightmare group.
- Discussion
4.1. Main findings
To the best of our knowledge the present study is the first to examine EEG, cardiac and motor activity in nightmare sufferers during different sleep state transitions. We showed that NM in comparison with CTL subjects exhibited significantly enhanced alpha EEG activity during the transition from NREM to REM state. In addition, motor activity during pre- and post-REM periods, resembled the findings related to alpha power: although the effects regarding muscle atonia showed only a weak trend, they indicated attenuated muscular atonia (enhanced motor activity) in NM subjects during pre-REM periods in contrast to post-REM ones. Furthermore, measures of HRV indexing autonomic regulation on cardiac activity showed significantly reduced values in the NM group in comparison with that of CTL subjects. While CTL subjects showed increased HRV during pre-REM periods in contrast to postREM ones, NM subjects did not exhibit such sleep state-specific differences in HRV, but showed more stable and reduced values of HRV reflecting generally attenuated parasympathetic activity. And finally, these differences were independent of the confounding effects of waking anxiety, suggesting that cortical and peripheral measures of altered sleep physiology preceding REM periods are related to the condition of disturbed dreaming (Nightmare Disorder), regardless of the confounding effects of co-morbid anxious symptoms.
4.2. The heterogeneity of NREM sleep
The separate analysis of sleep-stage transitions reveals the constraints of defining discrete ‘‘borders’’ for continuously changing physiological patterns comprising the coexistence of different sleep-like and wake-like phenomena. Although pre-and postREM periods are usually analyzed together as both segments belong to NREM sleep, our data are in line with studies presenting a more dynamic view of the ultradian process exceeding the boundaries of traditional sleep stages (Halász & Bódizs, 2013; Merica & Fortune, 1997; Nielsen, 2000). The main effects of Condition (analyzing the data of both groups as a whole) showed that pre-REM periods, in sharp contrast to post-REM ones were characterized by enhanced alpha power and motor activation as well as a trend of increased parasympathetic autonomic activity. (The less robust effect of condition regarding autonomic activity is due to the fact that the sleep-state specific effects were only present in the control group.) These findings show that NREM to REM sleep state transitions involve the attenuation of arousal thresholds leading to higher levels of cortical and motor activations.
NREM–REM transitions are characterized by the attenuation of slow wave activity, marking the reduction of (deep) sleep-promoting influences and the increase of REM/wake promoting neural activity (Halász & Bódizs, 2013; Halász, Terzano, Parrino, & Bódizs, 2004). During these intermediate states the coexistence of sleep-like and wake-like neural oscillations are likely to occur (Mahowald et al., 2011; Nobili et al., 2011). In contrast, following REM periods, sleep-promoting activity and higher thresholds of arousability are reinstated (Halász & Bódizs, 2013). Our data also resemble earlier reports on the heterogeneity of Stage 2 sleep, showing that this sleep state is ‘‘more active’’, characterized by abrupt motor, neurohormonal and autonomic changes before REM sleep, but in contrast, becomes ‘‘quieter’’ when progressing toward deeper sleep states (Pilcher & Schulz, 1987). We propose that the separate analyses of pre-and post-REM periods provides a more accurate picture about the cortical and peripheral ‘‘events’’ that are associated to changes in environmental awareness and presumably to the intensity of mental experiences during sleep.
4.3. Arousal-related activity in NM subjects during pre-REM periods
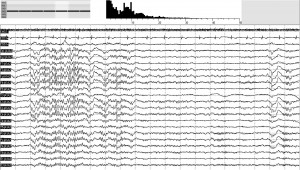
Our findings indicate that NM subjects’ sleep is altered and more activated during the transitions from NREM to REM sleep, the so-called ascending phases of sleep (Halász & Bódizs, 2013). Previous studies indicated that NM subjects’ sleep was characterized by impaired sleep continuity (Simor, Horváth, et al., 2012) and frequent microarousals (Simor, Bódizs, et al., 2013) during NREM, and enhanced high-alpha (10–14.5 Hz) power during REM periods (Simor, Horváth, et al., 2013); however, findings regarding alterations in NREM sleep were somewhat inconsistent: some studies did not find differences between NM subjects and paired controls in NREM and SWS sleep (Germain & Nielsen, 2003) while others reported reduced SWS and increased alpha activity during NREM sleep at a tendency level in NM subjects (Simor, Horváth, et al., 2012, 2013). Here, we complement and clarify these findings, by showing that NM sufferers’ sleep is particularly compromised during NREM–REM transitions. In these pre-REM periods, NM subjects compared to CTLs were characterized by enhanced alpha power, whereas during post-REM and stable, non-transitory NREM periods NMs did not exhibit significantly increased alpha power values. Fig. 4 illustrates a period of increased pre-REM alpha power in one of our nightmare subjects.
Although the conventional frequency range of alpha activity is defined between 8 and 13 Hz, the bin-wise analyses indicated that this increase was evident in an overlapping, but somewhat lower range, between 7 and 12.25 Hz. Enhanced alpha activity in the ‘‘proximity’’ of REM sleep is considered to reflect a ‘‘covert’’ wake-like state characterized by increased environmental awareness and higher arousability (Halász et al., 2004; McKinney et al., 2011; Parrino et al., 2012). Since EEG activity during post-REM periods was ‘‘reinstated’’ approaching values of the control group, we suggest that cortical activation reflected by enhanced alpha power in NM subjects may be triggered by physiological processes related to REM sleep pressure. Our findings resemble the assumptions of the covert REM sleep theory proposed by Nielsen (2000), claiming that physiological processes of REM sleep might appear out of the conventional borders of REM phases, especially during Stage 2 sleep before and after REM periods. Nevertheless, our findings indicate that NREM periods before are functionally different from NREM periods after REM sleep, and nightmare subjects are prone to exhibit EEG features reflecting a more alert brain state, specifically during sleep epochs that precede REM sleep.
Fig. 4. A 20-s segment of NREM–REM transition (pre-REM sleep) of a NM subjects’ polysomnographic recording. The coexistence of wake-like and sleep-like features is reflected by short period of alpha oscillations (on the left) and a K-complex and spindle activity (on the right), respectively. The hypnogram and the spectral power distribution of the segment are shown on the top of the figure.
A trend of enhanced activity in the frequency range of 17.75– 27.25 Hz during the pre-REM period was also found in NM in comparison to CTL subjects. Bin-wise spectral power analyses showed that the increase of a highly overlapping frequency range (18– 30 Hz) is specifically associated with the transition to REM from NREM sleep, and that fast frequency activity ranging from 18 Hz marks the division between NREM specific and REM/wake specific activity (Merica & Fortune, 2004). We propose that enhanced power of alpha and the tendency of increased higher (beta) frequency (17.75–27.25 Hz) activity during pre-REM periods in NM subjects indicates the augmentation of REM/wake promoting neural activity during NREM–REM transitions. Although the appearance of high frequency activity during such sleep state transitions is paralleled with the reduction of delta (1–4 Hz) activity (Merica & Fortune, 1997), NM subjects were not characterized by reduced power in the delta range during pre-or post-REM periods. This finding is in line with our assumption that during NREM–REM transitions, the coexistence of sleep-like and wake-like oscillations, as well as the proportion of sleep-promoting and REM/wake related neural influences may be considered as relevant neurophysiological features of Nightmare Disorder.
In addition to EEG measures, NMs showed a trend of relatively reduced muscle atonia during pre-REM in comparison to post-REM and non-transitory periods. Earlier studies reported increased motor activity in NMs during NREM and REM sleep (Germain & Nielsen, 2003). Our data resemble these findings and indicate that increased motor activity in NMs might preferentially take place during NREM–REM transitions.
And finally, we found reduced values of HRV indexed by the HF and the RMSSD measures, both reflecting reduced parasympathetic (vagal) control on cardiac activity in NM in comparison to that of CTL subjects. Significant differences emerged between pre-REM conditions, due to reduced parasympathetic regulation in the NM group irrespective of the specific sleep state. In contrast, the CTL group showed relatively increased HRV during pre-REM and reduced HRV during post-REM periods. Moreover, NMs were characterized by relatively reduced HRV (LF, HF, RMSSD) during nontransitory NREM periods, indicating generally impaired autonomic regulation during stabile and deeper NREM sleep states. While CTLs exhibited similar HRV values during post-REM and non-transitory NREM periods, NMs ‘‘approached’’ control values only during post-REM periods. To the best of our knowledge, only one previous study examined cardiac parameters in NM subjects, however their findings are difficult to compare to the present ones because of the differences in sample size, methodology and statistical procedures (Nielsen et al., 2010). Here, we showed that NMs are characterized by reduced HRV in pre-REM and non-transitory NREM periods which might indicate impaired cardiac regulation during NREM sleep. Since NMs showed reduced HRV during pre-REM periods, while CTLs showed relatively increased HRV values, we might also speculate that NM subjects, during episodes of enhanced cortical activity before the initiation of REM periods, fail to exhibit parasympathetic autonomic regulation on cardiac activity.
4.4. Arousal-related activity and oneiric experiences
Although empirical data on the relationship between arousals and mental activity during sleep is scarce, it is tempting to speculate that increased arousal-related activity in nightmare subjects during pre-REM periods is related to altered emotional and cognitive processes underlying the often intense and negatively toned dream experiences. Environmental stimulation inducing alpha activity was related to higher visual imagery reports during NREM sleep (Conduit et al., 1997) and spontaneous high frequency activity (10–12 and 14–18 Hz) was associated to successful dream recall from REM sleep (Chellappa, Frey, Knoblauch, & Cajochen, 2011). Moreover, subjects with high dream recall frequency showed more intra sleep wakefulness in a paradigm applying external stimulation (Ruby et al., 2013). In contrast, during a recovery night following sleep deprivation a marked impairment of NREM dream recall was found, paralleled by the attenuation of intra sleep wakefulness and micro-arousals (De Gennaro et al., 2010).
And finally, sleep paralysis, an unusual sleep phenomenon characterized by increased attention orienting toward external stimuli during REM sleep (Mahowald et al., 2011; Terzaghi et al., 2012) often involves perceptually intense, real-like and nightmarish dream experiences. Since the transition to REM sleep is related to the activation of a neural network comprising brainstem and several fronto-limbic structures (Abe, Ogawa, Nittono, & Hori, 2008; Muzur, Pace-Schott, & Hobson, 2002), we might suggest that wake-like cortical activity and lower parasympathetic control reflects enhanced emotional memory processing and impaired emotional regulation in nightmare sufferers (Levin & Nielsen, 2007). On the other hand, it is also plausible that arousals related to increased environmental awareness might intensify the perceptual aspects of mental experiences and facilitate the encoding of dreams.
4.5. Conclusions and future directions
In conclusion, we showed that NM subjects are characterized by enhanced wake-like cortical and slightly increased motor activity during NREM–REM transitions, as well as generally attenuated parasympathetic cardiac regulation. Our study argues for the heterogeneity of sleep, comprised by markedly different states reflecting the ascending and descending phases of sleep regulation. Within this theoretical framework, our data complements earlier findings examining the neurophysiological background of Nightmare Disorder, and provides empirical support for the role of REM sleep in triggering sleep disruptions in subjects with frequent nightmares. Future studies might address how the enhanced cortical activity paralleled with reduced parasympathetic control is associated with the emotional and perceptual aspects of nightmarish dream experiences. Since nightmares are intense emotional experiences we might speculate that cortical activations, enhanced muscle tone, and reduced parasympathetic control before the onset of REM sleep, are related to enhanced activity of limbic and motor structures processing emotional memories (Levin & Nielsen, 2007; Walker & van Der Helm, 2009) and/or behavioral patterns of flight/fight responses in subjects with frequent nightmares (Revonsuo, 2000). Nevertheless, although the altered neurophysiological patterns in NM subjects were not associated to waking anxiety, the nightmarish quality and negative emotionality of these dream experiences might be influenced by such psychological factors (Levin & Nielsen, 2007; Levin et al., 2011). Since the present work is the first to examine neurophysiological parameters in NM subjects during sleep state transitions, further studies with larger sample sizes and possibly more heterogeneous samples are required to support or challenge these preliminary findings. Moreover, although we controlled in our NM group for the confounding effects of waking anxiety, more research is warranted to examine the influence of more severe, co-morbid mental symptoms on neurophysiological measures during sleep.
Conflict of interest
None declared.
Funding
The funding source had no involvement in any part (study design, analyses and interpretation of data, etc.) of the study.
Acknowledgments
This research was supported by the European Union and the State of Hungary, co-financed by the European Social Fund in the framework of TÁMOP 4.2.4. A/-11-1-2012-0001 ‘National Excellence Program’.
References
Abe, T., Ogawa, K., Nittono, H., & Hori, T. (2008). Neural generators of brain potentials before rapid eye movements during human REM sleep: A study using sLORETA. Clinical Neurophysiology: Official Journal of the International Federation of Clinical Neurophysiology, 119, 2044–2053.
Abt, K. (1988). Descriptive data analysis (DDA) in quantitative EEG studies. Statistics and Topography in Quantitative EEG, 150–160.
American Academy of Sleep Medicine (2005). The international classification of sleep disorders: Diagnostic and coding manual (2nd ed.). Westchester, IL: American Academy of Sleep Medicine.
Benjamini, Y., Drai, D., Elmer, G., Kafkafi, N., & Golani, I. (2001). Controlling the false discovery rate in behavior genetics research. Behavioural Brain Research, 125, 279–284.
Bódizs, R., Simor, P., Csóka, S., Bérdi, M., & Kopp, M. S. (2008). Dreaming and health promotion: A theoretical proposal and some epidemiological establishments. European Journal of Mental Health, 3, 35–62.
Bódizs, R., Sverteczki, M., & Mészáros, E. (2008). Wakefulness–sleep transition: Emerging electroencephalographic similarities with the rapid eye movement phase. Brain Research Bulletin, 76, 85–89.
Chang, H.-A., Chang, C.-C., Tzeng, N.-S., Kuo, T. B., Lu, R.-B., & Huang, S.-Y. (2013). Decreased cardiac vagal control in drug-naïve patients with posttraumatic stress disorder. Psychiatry Investigation, 10, 121–130.
Chellappa, S. L., Frey, S., Knoblauch, V., & Cajochen, C. (2011). Cortical activation patterns herald successful dream recall after NREM and REM sleep. Biological Psychology, 87, 251–256.
Conduit, R., Bruck, D., & Coleman, G. (1997). Induction of visual imagery during NREM sleep. Sleep, 20, 948–956.
Coolidge, F. L., Segal, D. L., Coolidge, C. M., Spinath, F. M., & Gottschling, J. (2010). Do nightmares and generalized anxiety disorder in childhood and adolescence have a common genetic origin? Behavior Genetics, 40, 349–356.
De Gennaro, L., Ferrara, M., Curcio, G., & Cristiani, R. (2001). Antero-posterior EEG changes during the wakefulness–sleep transition. Clinical Neurophysiology: Official Journal of the International Federation of Clinical Neurophysiology, 112, 1901–1911.
De Gennaro, L., Marzano, C., Moroni, F., Curcio, G., Ferrara, M., & Cipolli, C. (2010). Recovery sleep after sleep deprivation almost completely abolishes dream recall. Behavioural Brain Research, 206, 293–298.
Dishman, R. K., Nakamura, Y., Garcia, M. E., Thompson, R. W., Dunn, A. L., & Blair, S. N. (2000). Heart rate variability, trait anxiety, and perceived stress among physically fit men and women. International Journal of Psychophysiology: Official Journal of the International Organization of Psychophysiology, 37, 121–133.
Duffy, F. H., Jones, K., Bartels, P., Albert, M., McAnulty, G. B., & Als, H. (1990). Quantified neurophysiology with mapping: Statistical inference, exploratory and confirmatory data analysis. Brain Topography, 3, 3–12.
Elsenbruch, S., Harnish, M. J., & Orr, W. C. (1999). Heart rate variability during waking and sleep in healthy males and females. Sleep, 22, 1067–1071.
Ferini-Strambi, L., Bianchi, A., Zucconi, M., Oldani, A., Castronovo, V., & Smirne, S. (2000). The impact of cyclic alternating pattern on heart rate variability during sleep in healthy young adults. Clinical Neurophysiology: Official Journal of the International Federation of Clinical Neurophysiology, 111, 99–101.
Ferri, R., Bruni, O., Fulda, S., Zucconi, M., & Plazzi, G. (2012). A quantitative analysis of the submentalis muscle electromyographic amplitude during rapid eye movement sleep across the lifespan. Journal of Sleep Research, 21, 257–263.
Ferri, R., Rundo, F., Manconi, M., Plazzi, G., Bruni, O., Oldani, A., et al. (2010). Improved computation of the atonia index in normal controls and patients with REM sleep behavior disorder. Sleep Medicine, 11, 947–949.
Germain, A., & Nielsen, T. A. (2003). Sleep pathophysiology in posttraumatic stress disorder and idiopathic nightmare sufferers. Biological Psychiatry, 54, 1092–1098.
Gorman, J. M., & Sloan, R. P. (2000). Heart rate variability in depressive and anxiety disorders. American Heart Journal, 140, 77–83.
Grigg-Damberger, M., Gozal, D., Marcus, C. L., Quan, S. F., Rosen, C. L., Chervin, R. D., et al. (2007). The visual scoring of sleep and arousal in infants and children. Journal of Clinical Sleep Medicine: JCSM: Official Publication of the American Academy of Sleep Medicine, 3(2), 201–240.
Halász, P., & Bódizs, R. (2013). Dynamic structure of NREM sleep. London: SpringerVerlag.
Halász, P., Terzano, M., Parrino, L., & Bódizs, R. (2004). The nature of arousal in sleep. Journal of Sleep Research, 13, 1–23. Heart Rate Variability (1996). Standards of measurement, physiological interpretation, and clinical use. Circulation, 93, 1043–1065.
Iber, C., Ancoli-Israel, S., Chesson, A., & Quan, S. F. (2007). The AASM manual for the scoring of sleep and associated events: Rules, terminology and technical specifications. Westchester, IL: American Academy of Sleep Medicine.
Jasper, H. (1958). Report of the committee on methods of clinical examination in electroencephalography. Electroencephalography and Clinical Neurophysiology, 10, 370–375.
Kemp, A. H., Quintana, D. S., Felmingham, K. L., Matthews, S., & Jelinek, H. F. (2012). Depression, comorbid anxiety disorders, and heart rate variability in physically healthy, unmedicated patients: Implications for cardiovascular risk. PLoS One, 7, e30777. http://dx.doi.org/10.1371/journal.pone.0030777.
Lancee, J., Spoormaker, V. I., & Van den Bout, J. (2010). Nightmare frequency is associated with subjective sleep quality but not with psychopathology. Sleep Biological Rhythms, 8, 187–193.
Levin, R., Fireman, G., Spendlove, S., & Pope, A. (2011). The relative contribution of affect load and affect distress as predictors of disturbed dreaming. Behavioral Sleep Medicine, 9, 173–183.
Levin, R., & Nielsen, T. A. (2007). Disturbed dreaming, posttraumatic stress disorder, and affect distress: A review and neurocognitive model. Psychological Bulletin, 133, 482–528.
Mahowald, M. W., Cramer Bornemann, M. A., & Schenck, C. H. (2011). State dissociation, human behavior, and consciousness. Current Topics in Medicinal Chemistry, 11, 2392–2402.
Manni, R., Terzaghi, M., & Glorioso, M. (2009). Motor-behavioral episodes in REM sleep behavior disorder and phasic events during REM sleep. Sleep, 32, 241–245. McKinney, S. M., Dang-Vu, T. T., Buxton, O. M., Solet, J. M., & Ellenbogen, J. M. (2011). Covert waking brain activity reveals instantaneous sleep depth. PLoS One, 6, e17351. Merica, H., & Fortune, R. D. (1997). A neuronal transition probability model for the evolution of power in the sigma and delta frequency bands of sleep EEG. Physiology & Behavior, 62, 585–589.
Merica, H., & Fortune, R. D. (2004). State transitions between wake and sleep, and within the ultradian cycle, with focus on the link to neuronal activity. Sleep Medicine Reviews, 8, 473–485.
Mizuki, Y., Kajimura, N., Nishikori, S., Imaizumi, J., & Yamada, M. (1984). Appearance of frontal midline theta rhythm and personality traits. Folia Psychiatrica et Neurologica Japonica, 38, 451–458.
Muzur, A., Pace-Schott, E. F., & Hobson, J. A. (2002). The prefrontal cortex in sleep. Trends in Cognitive Sciences, 6, 475–481. Nielsen, T. A. (2000). A review of mentation in REM and NREM sleep: ‘‘Covert’’ REM sleep as a possible reconciliation of two opposing models. Behavioral and Brain Sciences, 23, 851–866 (discussion 904–1121).
Nielsen, T., Paquette, T., Solomonova, E., Lara-Carrasco, J., Colombo, R., & Lanfranchi, P. (2010). Changes in cardiac variability after REM sleep deprivation in recurrent nightmares. Sleep, 33, 113–122.
Nobili, L., Ferrara, M., Moroni, F., De Gennaro, L., Russo, G. L., Campus, C., et al. (2011). Dissociated wake-like and sleep-like electro-cortical activity during sleep. NeuroImage, 58, 612–619.
Parrino, L., Ferri, R., Bruni, O., & Terzano, M. G. (2012). Cyclic alternating pattern (CAP): The marker of sleep instability. Sleep Medicine Reviews, 16, 27–45. Pilcher, J. J., & Schulz, H. (1987). The interaction between EEG and transient muscle activity during sleep in humans. Human Neurobiology, 6(1), 45–49.
Revonsuo, A. (2000). The reinterpretation of dreams: An evolutionary hypothesis of the function of dreaming. Behavioral and Brain Sciences, 23(6), 877–901.
Reyes del Paso, G. A., Langewitz, W., Mulder, L. J. M., van Roon, A., & Duschek, S. (2013). The utility of low frequency heart rate variability as an index of sympathetic cardiac tone: A review with emphasis on a reanalysis of previous studies. Psychophysiology, 50, 477–487.
Robert, G., & Zadra, A. (2014). Thematic and content analysis of idiopathic nightmares and bad dreams. Sleep, 37, 409–417.
Ruby, P., Blochet, C., Eichenlaub, J.-B., Bertrand, O., Morlet, D., & Bidet-Caulet, A. (2013). Alpha reactivity to first names differs in subjects with high and low dream recall frequency. Frontiers in Psychology, 4, 419.
Saper, C. B., Chou, T. C., & Scammell, T. E. (2001). The sleep switch: Hypothalamic control of sleep and wakefulness. Trends in Neurosciences, 24, 726–731.
Schredl, M. (2003). Effects of state and trait factors on nightmare frequency. European Archives of Psychiatry and Clinical Neuroscience, 253, 241–247.
Schredl, M. (2009). Nightmare frequency in patients with primary insomnia. International Journal of Dream Research, 2, 85–88. Schredl, M., Schafer, G., Weber, B., & Heuser, I. (1998). Dreaming and insomnia: Dream recall and dream content of patients with insomnia. Journal of Sleep Research, 7, 191–198.
Simor, P., Bódizs, R., Horváth, K., & Ferri, R. (2013). Disturbed dreaming and the instability of sleep: Altered nonrapid eye movement sleep microstructure in individuals with frequent nightmares as revealed by the cyclic alternating pattern. Sleep, 36, 413–419.
Simor, P., Horváth, K., Gombos, F., Takács, K. P., & Bódizs, R. (2012). Disturbed dreaming and sleep quality: Altered sleep architecture in subjects with frequent nightmares. European Archives of Psychiatry and Clinical Neuroscience, 262, 687–696.
Simor, P., Horváth, K., Ujma, P. P., Gombos, F., & Bódizs, R. (2013). Fluctuations between sleep and wakefulness: Wake-like features indicated by increased EEG alpha power during different sleep stages in nightmare disorder. Biological Psychology, 94, 592–600.
Simor, P., Kovács, I., Vargha, A., Csóka, S., Mangel, B., & Bódizs, R. (2009). Nightmares, dream anxiety and psychopathology: The validation of the Hungarian version of the Van Anxiety Scale. Psychiatria Hungarica, 24, 428–438.
Simor, P., Pajkossy, P., Horváth, K., & Bódizs, R. (2012). Impaired executive functions in subjects with frequent nightmares as reflected by performance in different neuropsychological tasks. Brain and Cognition, 78, 274–283.
Sipos, K., Sipos, M., & Spielberger, C. (1994). [The Hungarian version of the statetrait anxiety inventory (STAI)]. In F. Mérei & F. Szakács (Eds.) Pszichodiagnosztikai Vademecum. I/2 (pp. 123–148). Budapest: Nemzetközi Tankönyvkiadó.
Sjöström, N., Hetta, J., & Waern, M. (2009). Persistent nightmares are associated with repeat suicide attempt: A prospective study. Psychiatry Research, 170, 208–211.
Spiegelhalder, K., Fuchs, L., Ladwig, J., Kyle, S. D., Nissen, C., Voderholzer, U., et al. (2011). Heart rate and heart rate variability in subjectively reported insomnia. Journal of Sleep Research, 20, 137–145.
Spoormaker, V. I., Schredl, M., & van den Bout, J. (2006). Nightmares: From anxiety symptom to sleep disorder. Sleep Medicine Reviews, 10, 19–31.
Suetsugi, M., Mizuki, Y., Ushijima, I., Yamada, M., & Imaizumi, J. (1998). Anxiolytic effects of low-dose clomipramine in highly anxious healthy volunteers assessed by frontal midline theta activity. Progress in Neuro-Psychopharmacology and Biological Psychiatry, 22, 97–112.
Swart, M. L., van Schagen, A. M., Lancee, J., & van den Bout, J. (2013). Prevalence of nightmare disorder in psychiatric outpatients. Psychotherapy and Psychosomatics, 82, 267–268.
Terzaghi, M., Ratti, P. L., Manni, F., & Manni, R. (2012). Sleep paralysis in narcolepsy: More than just a motor dissociative phenomenon? Neurological Sciences: Official Journal of the Italian Neurological Society and of the Italian Society of Clinical Neurophysiology, 33, 169–172.
Terzaghi, M., Sartori, I., Tassi, L., Didato, G., Rustioni, V., LoRusso, G., et al. (2009). Evidence of dissociated arousal states during NREM parasomnia from an intracerebral neurophysiological study. Sleep, 32, 409–412.
Tyson, P. D., Ogilvie, R. D., & Hunt, H. T. (1984). Lucid, prelucid, and nonlucid dreams related to the amount of EEG alpha activity during REM sleep. Psychophysiology, 21, 442–451.
Van Liempt, S., van Zuiden, M., Westenberg, H., Super, A., & Vermetten, E. (2013). Impact of impaired sleep on the development of PTSD symptoms in combat veterans: A prospective longitudinal cohort study. Depression and Anxiety, 30, 469–474.
Vgontzas, A. N., Fernandez-Mendoza, J., Liao, D., & Bixler, E. O. (2013). Insomnia with objective short sleep duration: The most biologically severe phenotype of the disorder. Sleep Medicine Reviews, 17, 241–254.
Voss, U., Holzmann, R., Tuin, I., & Hobson, J. A. (2009). Lucid dreaming: A state of consciousness with features of both waking and non-lucid dreaming. Sleep, 32, 1191–1200. Walker, M. P., & van Der Helm, E. (2009). Overnight therapy? The role of sleep in emotional brain processing. Psychological Bulletin, 135, 731–748.
Willmann, M., Langlet, C., Hainaut, J.-P., & Bolmont, B. (2012). The time course of autonomic parameters and muscle tension during recovery following a moderate cognitive stressor: Dependency on trait anxiety level. International Journal of Psychophysiology: Official Journal of the International Organization of Psychophysiology, 84, 51–58.
Wood, J. M., & Bootzin, R. R. (1990). The prevalence of nightmares and their independence from anxiety. Journal of Abnormal Psychology, 99, 64–68.
Zadra, A., Pilon, M., & Donderi, D. C. (2006). Variety and intensity of emotions in nightmares and bad dreams. Journal of Nervous and Mental Disease, 194, 249–254.