Research > Topics > A parahippocampal electrocorticographic analysis of the transition from wakefulness to sleep in humans: are REM features present? (2002 – 2004, European Sleep Research Society – Sanofi-Synthelabo Grant)
The aim of the current study is to test a physiological prediction of the covert REM sleep hypothesis of dreaming. According to the above mentioned hypothesis “hidden” elements of REM physiology are present during sleep onset explaining the relatively vivid dreaming during this transitional period (Nielsen, 2000).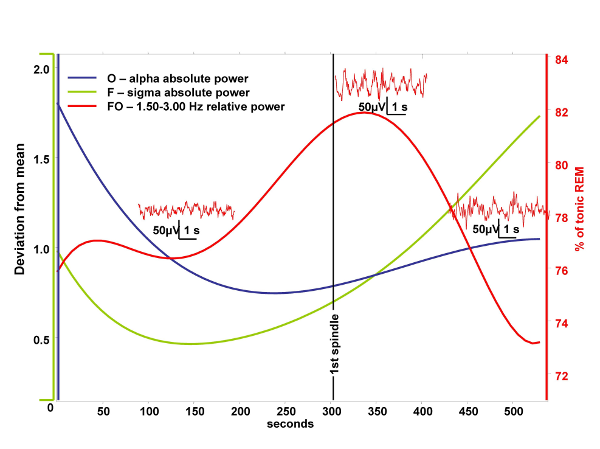 The temporal resolution of functional neuroimaging techniques is too low to study the transition between wakefulness and sleep. In turn parahippocampal electrocorticography based on foramen ovale electrodes has the advantage of a high temporal resolution combined with the ability to measure mediotemporal population activity in a semi-invasive way. Previously we found a high REM-specificity of 1.50-3.00 Hz relative power in foramen ovale recordings (Bódizs et al, 2001). We suppose that this oscillation regularly appears during the whole stage 1 and the initial part of stage 2 sleep and negatively correlates with occipitally recorded alpha activity of the electroencephalogram. Our study is based on foramen ovale recordings of epileptic patients undergoing presurgical evaluation. We selected 9 minutes long records containing the last 5 minutes before and the first 4 minutes after the first visually detected scalp-EEG spindle. Evolution of 0.25-1.25 Hz, 1.50-3.00 Hz, 3.25-4.50 Hz, 4.75-6.25 Hz, 6.50-7.75 Hz, 8-12 Hz (alpha), 12.25-15 Hz (sigma), 15.25-30 Hz (beta) absolute and relative power were assessed by Fast Fourier Transformation (FFT) of consecutive 4 second epochs (50% overlap, Hanning-window). Power values of adjacent 4 epochs (10 s) were averaged. An additional smoothing consisting of a 3 points moving average was performed. We selected 30-50 artifact- and spike-free 4 s epochs from tonic REM sleep and normalized the 1.50-3.00 Hz relative power of the parahippocampal wakefulness-sleep transition signal by expressing it as a percent of the individual 1.50-3.00 Hz relative power of tonic REM sleep. A parahippocampal channel from those which are contralateral to the ictal pacemaker as well as the frontal (F3 or F4), temporal (T3 or T4) and occipital (O1 or O2) channels of the same side (all vertex-referred) were selected for analysis. In those cases in which bitemporal epilepsy was diagnosed we chose the least affected parahippocampal channel for analysis. A least squares 5th order polynomial fit of the temporal evolution of occipital alpha and frontal sigma power as well as that of the 1.50-3.00 Hz parahippocampal activity was performed and the results were averaged between subjects. This resulted in falling and rising trends of alpha and sigma powers, respectively and in a slightly skewed, bell-shaped curve expressing the evolution of 1.50-3.00 Hz parahippocampal activity. The peak of the latter fell between the periods of alpha and sigma dominance averagely 30 seconds after the first sleep spindle was observed and it reached 82% of tonic REM sleep values (Fig. 1.). Skewness resulted from the slower increase of 1.50-3.00 Hz activity during the period of alpha dropout. The mean correlation between occipital alpha power and parahippocampal 1.50-3.00 Hz REM-like activity was r = -0.13, which does not reach the level of significance. There is no correlation between frontal sigma power and parahippocampal REM-like activity (r = 0.08). The temporal resolution of functional neuroimaging techniques is too low to study the transition between wakefulness and sleep. In turn parahippocampal electrocorticography based on foramen ovale electrodes has the advantage of a high temporal resolution combined with the ability to measure mediotemporal population activity in a semi-invasive way. Previously we found a high REM-specificity of 1.50-3.00 Hz relative power in foramen ovale recordings (Bódizs et al, 2001). We suppose that this oscillation regularly appears during the whole stage 1 and the initial part of stage 2 sleep and negatively correlates with occipitally recorded alpha activity of the electroencephalogram. Our study is based on foramen ovale recordings of epileptic patients undergoing presurgical evaluation. We selected 9 minutes long records containing the last 5 minutes before and the first 4 minutes after the first visually detected scalp-EEG spindle. Evolution of 0.25-1.25 Hz, 1.50-3.00 Hz, 3.25-4.50 Hz, 4.75-6.25 Hz, 6.50-7.75 Hz, 8-12 Hz (alpha), 12.25-15 Hz (sigma), 15.25-30 Hz (beta) absolute and relative power were assessed by Fast Fourier Transformation (FFT) of consecutive 4 second epochs (50% overlap, Hanning-window). Power values of adjacent 4 epochs (10 s) were averaged. An additional smoothing consisting of a 3 points moving average was performed. We selected 30-50 artifact- and spike-free 4 s epochs from tonic REM sleep and normalized the 1.50-3.00 Hz relative power of the parahippocampal wakefulness-sleep transition signal by expressing it as a percent of the individual 1.50-3.00 Hz relative power of tonic REM sleep. A parahippocampal channel from those which are contralateral to the ictal pacemaker as well as the frontal (F3 or F4), temporal (T3 or T4) and occipital (O1 or O2) channels of the same side (all vertex-referred) were selected for analysis. In those cases in which bitemporal epilepsy was diagnosed we chose the least affected parahippocampal channel for analysis. A least squares 5th order polynomial fit of the temporal evolution of occipital alpha and frontal sigma power as well as that of the 1.50-3.00 Hz parahippocampal activity was performed and the results were averaged between subjects. This resulted in falling and rising trends of alpha and sigma powers, respectively and in a slightly skewed, bell-shaped curve expressing the evolution of 1.50-3.00 Hz parahippocampal activity. The peak of the latter fell between the periods of alpha and sigma dominance averagely 30 seconds after the first sleep spindle was observed and it reached 82% of tonic REM sleep values (Fig. 1.). Skewness resulted from the slower increase of 1.50-3.00 Hz activity during the period of alpha dropout. The mean correlation between occipital alpha power and parahippocampal 1.50-3.00 Hz REM-like activity was r = -0.13, which does not reach the level of significance. There is no correlation between frontal sigma power and parahippocampal REM-like activity (r = 0.08).
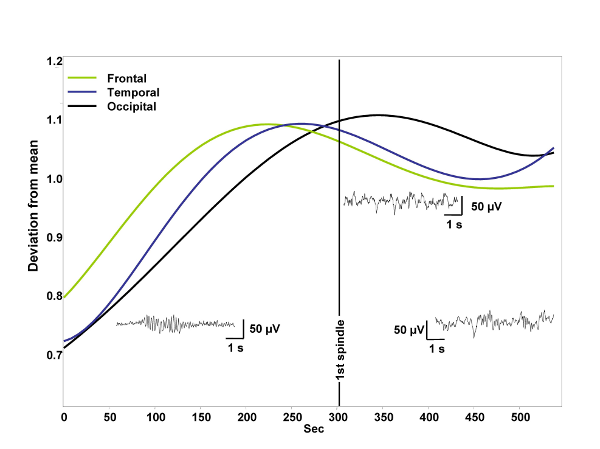 The 0.25-1.25 Hz relative power increased linearly in all channels. This does not correlate with the 1.50-3.00 Hz relative power, and it does not explain the latter’s temporal evolution. Relative power of all frequency bands above 3 Hz increased, peaked and decreased consistently earlier than 1.50-3.00 Hz activity in all channels. The only exception was beta power, which had been at his highest level already at the beginning and thereafter it decreased during the whole period. Absolute power values of all frequency bands except that of alpha and beta increased in all channels during the wakefulness-sleep transition. Absolute alpha and absolute beta power decreased. No other frequency band showed the bell shaped curve peaking shortly after the middle of the transitional period observed in 1.50-3.00 Hz relative power of the parahippocampal electrocorticogram. Indeed scalp-EEG recorded 1.50-3.00 Hz relative power showed similar, but less consistent peaks in all three channels. There was an anterior-posterior gradient in the time of peaking: frontal 1.50-3.00 Hz relative power peaked earlier than temporal and temporal earlier than occipital. The latter resembled considerably the parahippocampal REM-like activity’s trend (Fig. 2.). The 0.25-1.25 Hz relative power increased linearly in all channels. This does not correlate with the 1.50-3.00 Hz relative power, and it does not explain the latter’s temporal evolution. Relative power of all frequency bands above 3 Hz increased, peaked and decreased consistently earlier than 1.50-3.00 Hz activity in all channels. The only exception was beta power, which had been at his highest level already at the beginning and thereafter it decreased during the whole period. Absolute power values of all frequency bands except that of alpha and beta increased in all channels during the wakefulness-sleep transition. Absolute alpha and absolute beta power decreased. No other frequency band showed the bell shaped curve peaking shortly after the middle of the transitional period observed in 1.50-3.00 Hz relative power of the parahippocampal electrocorticogram. Indeed scalp-EEG recorded 1.50-3.00 Hz relative power showed similar, but less consistent peaks in all three channels. There was an anterior-posterior gradient in the time of peaking: frontal 1.50-3.00 Hz relative power peaked earlier than temporal and temporal earlier than occipital. The latter resembled considerably the parahippocampal REM-like activity’s trend (Fig. 2.).
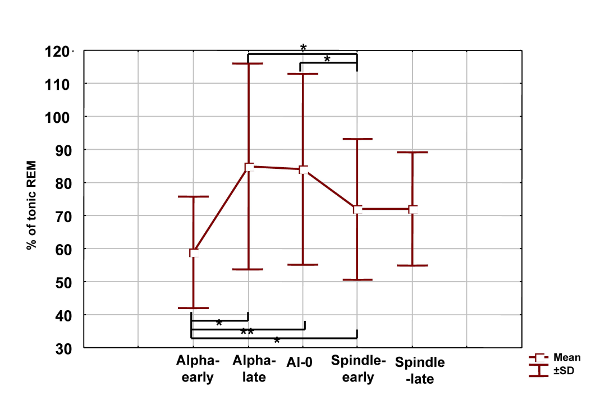
In the next step we intended to compare the wakefulness-sleep transition period with more stable states of consciousness. Thirty to fifty artifact and spike-free 4 s epochs dominated by alpha waves or stage 2 sleep, but outside from the wakefulness-sleep transition period were selected and analyzed by FFT. Moreover, epochs of the wakefulness-sleep transition period were visually categorized in alpha, alpha dropout and spindle/stage 2 states. The individually normalized 1.50-3.00 Hz relative power during stable wakefulness (alpha-early) was significantly lower, than during either of the transitional states, including the alpha-dominated epochs (alpha-late). However it does not differed significantly from stable stage 2 sleep (spindle-late). Alpha-dominated transitional periods (alpha-late) and alpha-dropout transitional periods (alpha-0) did not differ significantly, but both of them showed higher 1.50-3.00 Hz activity than early-spindling (Fig. 3.).
Taking into consideration the low non-significant negative correlation between occipital alpha power and parahippocampal 1.50-3.00 Hz activity (see above) the obtained results suggest that alpha waves are not related to the REM-like parahippocampal activity. It seems that just approaching sleep onset per se has a 1.50-3.00 Hz REM-like parahippocampal activity-inducing property.
 Results partially confirmed our hypotheses. REM-like 1.50-3.00 Hz activity increases during the transition from wakefulness to sleep, reaching averagely 82% of tonic REM. However, this REM-like physiological feature seems not to depend on alpha activity. The REM-like parahippocampal activity observed during wakefulness-sleep transition in this study harmonizes with the covert REM sleep hypothesis of dreaming (Nielsen, 2000) and with data regarding the lack of hippocampal involvement in hypnagogic imagery (Stickgold et al, 2000). The reason of the lack of involvement could be the fact that hippocampal output is consistently low during rhythmic slow activity (Buzsáki, 1996). Our findings regarding the temporal evolution of absolute power values are in accordance with EEG-studies focusing on sleep onset (De Gennaro et al, 2001), and complete them with the results of parahippocampal electrocorticographic analysis. Results partially confirmed our hypotheses. REM-like 1.50-3.00 Hz activity increases during the transition from wakefulness to sleep, reaching averagely 82% of tonic REM. However, this REM-like physiological feature seems not to depend on alpha activity. The REM-like parahippocampal activity observed during wakefulness-sleep transition in this study harmonizes with the covert REM sleep hypothesis of dreaming (Nielsen, 2000) and with data regarding the lack of hippocampal involvement in hypnagogic imagery (Stickgold et al, 2000). The reason of the lack of involvement could be the fact that hippocampal output is consistently low during rhythmic slow activity (Buzsáki, 1996). Our findings regarding the temporal evolution of absolute power values are in accordance with EEG-studies focusing on sleep onset (De Gennaro et al, 2001), and complete them with the results of parahippocampal electrocorticographic analysis.
References
- Bódizs, R., Kántor, S., Szabó, G., Szűcs, A., Erőss, L., Halász, P. (2001). Rhythmic hippocampal slow oscillation characterizes REM sleep in humans, Hippocampus, 11(6):747-753.
- Buzsáki, G. (1996). The hippocampo-neocortical dialogue. Cereb Cortex, 6:81-92.
- De Gennaro, L., Ferrara, M., Bertini, M. (2001). The boundary between wakefulness and sleep: quantitative electroencephalographic changes during the sleep onset period. Neuroscience, 107(1):1-11.
- Nielsen, T.A. (2000). Mentation in REM and NREM sleep: A review and possible reconciliation of two models. Behav Brain Sci, 23(6):851-66.
- Stickgold, R., Malia, A., Maguire, D., Roddenberry, D., O’Connor, M. (2000). Replaying the game: hypnagogic images in normals and amnesiacs. Science 290:350–3.
*Research supported by the ESRS – Sanofi-Synthelabo Grant 2002. ¶ See other details in Bódizs R et al.Brain Res Bull 2005; 65: 169-176. |
 The temporal resolution of functional neuroimaging techniques is too low to study the transition between wakefulness and sleep. In turn parahippocampal electrocorticography based on foramen ovale electrodes has the advantage of a high temporal resolution combined with the ability to measure mediotemporal population activity in a semi-invasive way. Previously we found a high REM-specificity of 1.50-3.00 Hz relative power in foramen ovale recordings (Bódizs et al, 2001). We suppose that this oscillation regularly appears during the whole stage 1 and the initial part of stage 2 sleep and negatively correlates with occipitally recorded alpha activity of the electroencephalogram. Our study is based on foramen ovale recordings of epileptic patients undergoing presurgical evaluation. We selected 9 minutes long records containing the last 5 minutes before and the first 4 minutes after the first visually detected scalp-EEG spindle. Evolution of 0.25-1.25 Hz, 1.50-3.00 Hz, 3.25-4.50 Hz, 4.75-6.25 Hz, 6.50-7.75 Hz, 8-12 Hz (alpha), 12.25-15 Hz (sigma), 15.25-30 Hz (beta) absolute and relative power were assessed by Fast Fourier Transformation (FFT) of consecutive 4 second epochs (50% overlap, Hanning-window). Power values of adjacent 4 epochs (10 s) were averaged. An additional smoothing consisting of a 3 points moving average was performed. We selected 30-50 artifact- and spike-free 4 s epochs from tonic REM sleep and normalized the 1.50-3.00 Hz relative power of the parahippocampal wakefulness-sleep transition signal by expressing it as a percent of the individual 1.50-3.00 Hz relative power of tonic REM sleep. A parahippocampal channel from those which are contralateral to the ictal pacemaker as well as the frontal (F3 or F4), temporal (T3 or T4) and occipital (O1 or O2) channels of the same side (all vertex-referred) were selected for analysis. In those cases in which bitemporal epilepsy was diagnosed we chose the least affected parahippocampal channel for analysis. A least squares 5th order polynomial fit of the temporal evolution of occipital alpha and frontal sigma power as well as that of the 1.50-3.00 Hz parahippocampal activity was performed and the results were averaged between subjects. This resulted in falling and rising trends of alpha and sigma powers, respectively and in a slightly skewed, bell-shaped curve expressing the evolution of 1.50-3.00 Hz parahippocampal activity. The peak of the latter fell between the periods of alpha and sigma dominance averagely 30 seconds after the first sleep spindle was observed and it reached 82% of tonic REM sleep values (Fig. 1.). Skewness resulted from the slower increase of 1.50-3.00 Hz activity during the period of alpha dropout. The mean correlation between occipital alpha power and parahippocampal 1.50-3.00 Hz REM-like activity was r = -0.13, which does not reach the level of significance. There is no correlation between frontal sigma power and parahippocampal REM-like activity (r = 0.08).
The temporal resolution of functional neuroimaging techniques is too low to study the transition between wakefulness and sleep. In turn parahippocampal electrocorticography based on foramen ovale electrodes has the advantage of a high temporal resolution combined with the ability to measure mediotemporal population activity in a semi-invasive way. Previously we found a high REM-specificity of 1.50-3.00 Hz relative power in foramen ovale recordings (Bódizs et al, 2001). We suppose that this oscillation regularly appears during the whole stage 1 and the initial part of stage 2 sleep and negatively correlates with occipitally recorded alpha activity of the electroencephalogram. Our study is based on foramen ovale recordings of epileptic patients undergoing presurgical evaluation. We selected 9 minutes long records containing the last 5 minutes before and the first 4 minutes after the first visually detected scalp-EEG spindle. Evolution of 0.25-1.25 Hz, 1.50-3.00 Hz, 3.25-4.50 Hz, 4.75-6.25 Hz, 6.50-7.75 Hz, 8-12 Hz (alpha), 12.25-15 Hz (sigma), 15.25-30 Hz (beta) absolute and relative power were assessed by Fast Fourier Transformation (FFT) of consecutive 4 second epochs (50% overlap, Hanning-window). Power values of adjacent 4 epochs (10 s) were averaged. An additional smoothing consisting of a 3 points moving average was performed. We selected 30-50 artifact- and spike-free 4 s epochs from tonic REM sleep and normalized the 1.50-3.00 Hz relative power of the parahippocampal wakefulness-sleep transition signal by expressing it as a percent of the individual 1.50-3.00 Hz relative power of tonic REM sleep. A parahippocampal channel from those which are contralateral to the ictal pacemaker as well as the frontal (F3 or F4), temporal (T3 or T4) and occipital (O1 or O2) channels of the same side (all vertex-referred) were selected for analysis. In those cases in which bitemporal epilepsy was diagnosed we chose the least affected parahippocampal channel for analysis. A least squares 5th order polynomial fit of the temporal evolution of occipital alpha and frontal sigma power as well as that of the 1.50-3.00 Hz parahippocampal activity was performed and the results were averaged between subjects. This resulted in falling and rising trends of alpha and sigma powers, respectively and in a slightly skewed, bell-shaped curve expressing the evolution of 1.50-3.00 Hz parahippocampal activity. The peak of the latter fell between the periods of alpha and sigma dominance averagely 30 seconds after the first sleep spindle was observed and it reached 82% of tonic REM sleep values (Fig. 1.). Skewness resulted from the slower increase of 1.50-3.00 Hz activity during the period of alpha dropout. The mean correlation between occipital alpha power and parahippocampal 1.50-3.00 Hz REM-like activity was r = -0.13, which does not reach the level of significance. There is no correlation between frontal sigma power and parahippocampal REM-like activity (r = 0.08). The 0.25-1.25 Hz relative power increased linearly in all channels. This does not correlate with the 1.50-3.00 Hz relative power, and it does not explain the latter’s temporal evolution. Relative power of all frequency bands above 3 Hz increased, peaked and decreased consistently earlier than 1.50-3.00 Hz activity in all channels. The only exception was beta power, which had been at his highest level already at the beginning and thereafter it decreased during the whole period. Absolute power values of all frequency bands except that of alpha and beta increased in all channels during the wakefulness-sleep transition. Absolute alpha and absolute beta power decreased. No other frequency band showed the bell shaped curve peaking shortly after the middle of the transitional period observed in 1.50-3.00 Hz relative power of the parahippocampal electrocorticogram. Indeed scalp-EEG recorded 1.50-3.00 Hz relative power showed similar, but less consistent peaks in all three channels. There was an anterior-posterior gradient in the time of peaking: frontal 1.50-3.00 Hz relative power peaked earlier than temporal and temporal earlier than occipital. The latter resembled considerably the parahippocampal REM-like activity’s trend (Fig. 2.).
The 0.25-1.25 Hz relative power increased linearly in all channels. This does not correlate with the 1.50-3.00 Hz relative power, and it does not explain the latter’s temporal evolution. Relative power of all frequency bands above 3 Hz increased, peaked and decreased consistently earlier than 1.50-3.00 Hz activity in all channels. The only exception was beta power, which had been at his highest level already at the beginning and thereafter it decreased during the whole period. Absolute power values of all frequency bands except that of alpha and beta increased in all channels during the wakefulness-sleep transition. Absolute alpha and absolute beta power decreased. No other frequency band showed the bell shaped curve peaking shortly after the middle of the transitional period observed in 1.50-3.00 Hz relative power of the parahippocampal electrocorticogram. Indeed scalp-EEG recorded 1.50-3.00 Hz relative power showed similar, but less consistent peaks in all three channels. There was an anterior-posterior gradient in the time of peaking: frontal 1.50-3.00 Hz relative power peaked earlier than temporal and temporal earlier than occipital. The latter resembled considerably the parahippocampal REM-like activity’s trend (Fig. 2.). Results partially confirmed our hypotheses. REM-like 1.50-3.00 Hz activity increases during the transition from wakefulness to sleep, reaching averagely 82% of tonic REM. However, this REM-like physiological feature seems not to depend on alpha activity. The REM-like parahippocampal activity observed during wakefulness-sleep transition in this study harmonizes with the covert REM sleep hypothesis of dreaming (Nielsen, 2000) and with data regarding the lack of hippocampal involvement in hypnagogic imagery (Stickgold et al, 2000). The reason of the lack of involvement could be the fact that hippocampal output is consistently low during rhythmic slow activity (Buzsáki, 1996). Our findings regarding the temporal evolution of absolute power values are in accordance with EEG-studies focusing on sleep onset (De Gennaro et al, 2001), and complete them with the results of parahippocampal electrocorticographic analysis.
Results partially confirmed our hypotheses. REM-like 1.50-3.00 Hz activity increases during the transition from wakefulness to sleep, reaching averagely 82% of tonic REM. However, this REM-like physiological feature seems not to depend on alpha activity. The REM-like parahippocampal activity observed during wakefulness-sleep transition in this study harmonizes with the covert REM sleep hypothesis of dreaming (Nielsen, 2000) and with data regarding the lack of hippocampal involvement in hypnagogic imagery (Stickgold et al, 2000). The reason of the lack of involvement could be the fact that hippocampal output is consistently low during rhythmic slow activity (Buzsáki, 1996). Our findings regarding the temporal evolution of absolute power values are in accordance with EEG-studies focusing on sleep onset (De Gennaro et al, 2001), and complete them with the results of parahippocampal electrocorticographic analysis.