Epilepsy as a derailment of sleep plastic functions may cause chronic cognitive impairment – a theoretical review
Sleep Medicine Reviews, vol. 45, June 2019, pages 31-41.
DOI: 10.1016/j.smrv.2019.01.003
Péter Halász1, Péter Ujma2, Dániel Fabó1, Róbert Bódizs2,1, Anna Szűcs1,2
1 National Institute of Clinical Neurosciences, Amerikai út 57. Budapest, H-1145, Hungary
2 Semmelweis University, Institute of Behavioral Sciences, Nagyvárad tér 4, Budapest, H-1089, Hungary
Summary
We report on a peculiar way of chronic cognitive impairment associated with interictal epileptic activity during NREM sleep. We review three major groups of epilepsy: mesiotemporal epilepsy (MTLE) involving the epileptic derailment of the hippocampal declarative memory system; childhood developmental epileptic encephalopathies; and the spectrum disorders of the perisylvian communication network with the centrotemporal spike phenomenon, overarching child- and adulthood epilepsies, totaling up the majority of epilepsies in childhood.
We outline high impact research-lines on the cognitive harm of epilepsy; causing specific or global cognitive decline through its interference with sleep plastic functions.
We highlight the key role of interictal activity in the development of cognitive impairment and the fact that we are unarmed against this harm, antiepileptic pharmaco-therapy being ineffective against the interictal process marked by spikes and high frequency oscillations.
Keywords:
Sleep-related epilepsies; cognitive harm; interictal epileptic activity; sleep slow waves; spindles; mesiotemporal epilepsy; epileptic encephalopathies; age dependent childhood focal epilepsies; centrotemporal spikes; Rolandic epilepsy
Practice Points
Due to the link of epilepsy and NREM sleep, sleep EEG is indispensable in the evaluation of epilepsies.
Sleep relatedness and interictal epileptic activity have major role in chronic cognitive harm.
Sleep EEG may clarify the cause of unexplained childhood cognitive decline or dysphasia.
Research Agenda
Assessing the share of spiking and structural hippocampal damage in the cognitive loss of MTLE.
Cognitive impairment needs to be correlated with the enhancement of sleep spiking in conditions with cognitive decline.
There is pressing need to find pharmaco-therapy treatment against interictal epileptic activity.
List of abbreviations
CA1, 2 Cornu Ammonis regions
CTS Centrotemporal spike
CAP Cyclic alternating pattern
ESES Electrical status epileptic in sleep
EE Epileptic encephalopathy
250-500 Hz Fast ripples
HFO (80–500 Hz) High frequency oscillations
IFCE Idiopathic focal childhood epilepsies = perisylvian network epilepsies
IED Interictal epileptic discharge
LKS Landau-Kleffner syndrome
LTP Long-term potentiation
MTLE Mesial temporal lobe epilepsy
PS Panayiotopoulos syndrome
HFO with 250-.500 Hz Pathological (epileptic) HFO
PN Perisylvian cognitive network
HFO with 100-250 Hz Physiological HFO
80 – 250 Hz oscillations Ripples
PN Perisylvian cognitive network
RE Rolandic epilepsy
SPW-R Sharp wave ripple complex
SW Spike wave pattern
TCI Transient cognitive impairment
Introduction
Cognitive impairment and epilepsy
The cognitive impairment of epileptic patients has multiple origins. In this review, we do not deal with structural brain lesions underlying epilepsies and disregard the effect antiepileptic drugs on cognitive functions, rather, our focus is the cognitive harm caused by epileptic disorder itself.
Epilepsy research has provided convincing proofs on the existence of postictal cognitive deficits depending on the duration, localization and extension of seizures [1]. The cognitive impairment related to interictal epileptic discharges (IEDs) is less clear [2].
First Colin Binnie raised and demonstrated in several papers the instant cognitive harm (transient cognitive impairment, TCI) of centrotemporal spikes (CTS) in Rolandic Epilepsy (RE) and of bilateral spike-waves (SW) in idiopathic generalized epilepsy [3,4]; making clinicians wonder whether the frequent spiking in idiopathic focal childhood epilepsies (IFCE) needed treatment for preventing cognitive decline. While RE patients’ transient cognitive signs apparently related to spiking supported this concept, the lack of cognitive impairment in their relatives carrying CTS made it ambiguous. The research on TCI had serious limitations. It assessed the instantaneous cognitive change associated to one IED only and, raised the chronic cognitive harm of IEDs just theoretically.
The coupling and interference of IEDs with plastic sleep functions shed special light on the chronic cognitive impairment associated to epilepsies [5]. These data suggest that beyond structural hippocampal sclerosis, an important functional factor – sleep-related spiking-contributes to the cognitive loss in mesio-temporal lobe epilepsy (MTLE).
Thence the link between memory disturbances and hippocampal spiking during sleep became clear in MTLE [6-8] and the association of cognitive decline with the sleep-activation of IEDs in epileptic encephalopathy children has been scrutinized [9-14]. Both lines of research provided strong evidence on the interference of IEDs with sleep plastic functions occurring during NREM sleep.
The most instrumental studies dealt with three major groups of epilepsies:
1) MTLE, the most frequent adulthood epilepsy;
2) The spectrum disorders of the perisylvian epileptic network (PN) constituted by the most frequent focal/regional age-dependent childhood epilepsies;
3) Epileptic encephalopathies including developmental childhood conditions.
We review below the mechanisms of cognitive impairment caused by IEDs in these groups of epilepsies.
The hippocampo-frontal dysfunction in MTLE impairs declarative memory
The hippocampo-frontal memory system strongly associates to NREM sleep. The active systems consolidation theory [13] states that memory traces initially stored in hippocampal circuits are transferred to neocortical networks via the interplay of NREM sleep-related electroencephalographic (EEG) components as slow waves, spindles and hippocampal sharp wave ripples (SPW-Rs).
SPW-R is a robust, far-reaching population episode, the most synchronized event of the mammalian brain [15], elevating the excitability of the hippocampus and connected structures [16-19]. It occurs in isolated hippocampal slices as well. The ripple is a high frequency (200Hz in animals, 150Hz in humans) event of the cornu ammonis (CA)1 pyramidal layer of the hippocampus, evoked by the sharp wave originating in CA3 (Figure 1.) [16]. It remains local between the CA3 pyramidal cells and the peri-somatic inhibitory neurons [20,21]. Its duration in rat is 30-150ms and the amplitude does not exceed 2.5mV. The spikes are smaller and shorter, and the coupled ripples are more synchronous in humans (Figure 2.). The big amount (50,000-150,000) of hippocampal neurons involved in SPW-R elicit long-term potentiation (LTP), which in turn changes the synaptic weights between the hippocampal neurons.
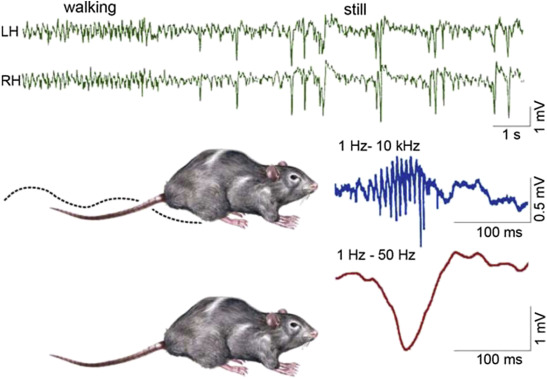
Figure 1.
Schematic picture of sharp-waves and ripples in rat hippocampus during the transition period from waking to sleep. Top: EEG during active waking state (locomotion and orientation): theta activity (left); during quiet wakefulness sharp-wave-ripple complexes appear (right). Bottom: the two states of the animal (A: activity, B: quiet wakefulness). (C) Enlarged picture of the sharp wave ripple complex: ripple (top) sharp wave (bottom) (After Buzsáki 2015).
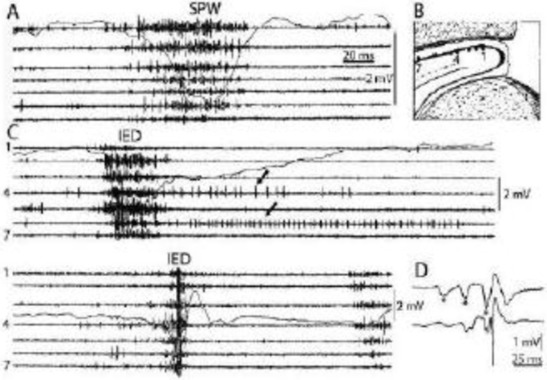
Figure 2.
Comparison of sharp waves (SPW) in the intact rat hippocampus (A) and interictal epileptic discharges (IEDs) after disconnection from its subcortical connections (C), by fimbria-fornix lesion (B). Channels represent different locations along the axis of the hippocampus (1-7). Arrows show prolonged post-IED activity in two putative interneurons. IEDs were the products of a reverberation in the entorhinal cortex-hippocampus loop evoked by entorhinal induced responses (asterisks) in insert (D) bottom trace represents the CA1 pyramidal layer Reverberation was terminated by the appearance of a large population spike. See tighter synchrony of population bursts and larger amplitude of the field responses during IEDs. (After Buzsáki 2015 and 1991).
The elimination of SPW-Rs abrogates memory consolidation [22,23]. Animal studies show that the cellular spike sequences in slow wave sleep are identical with the sequence found during SPW-Rs only faster and compressed [24-26]; the increase of the number of SPW-Rs after learning supports a similar scenario in humans [27,28].
The change of the hippocampo-neocortical memory process in MTLE
SPW-Rs fringe on the edge of a shift to epileptic excitation, giving a hint, why the hippocampus is the most epilepsy-prone structure of the brain. SPW-R is the normal counterpart of the epileptic twin, spike-fast ripple [15]. Ripples above 250 Hz are essential markers of epilepsy [29]. Also the epileptic spike is similar to SPW-R sharp waves just shorter, smaller and linking with high frequency oscillations (HFO) (fast ripples) (Figure 2). This similarity of the physiological and pathological variants highlights the vicinity of epilepsy to normal functioning. Stimulating the ventral commissure, Shatskikh et al. elicited large population spikes in the CA1 region of the hippocampal pyramidal neurons. The elicited spikes were similar to the spontaneous ones. The stimulated animals developed serious memory loss. Clemens et al. and Kleen et al. [7,8] evidenced the link between memory disturbances and hippocampal sleep spiking in human MTLE patients. Frauscher et al [30] studied hippocampal sleep spindling in human MTLE with implanted deep electrodes. Because hippocampal spindling and the increase of spiking correlated negatively, they assumed that spindles transformed to spikes; similarly to the proposed transformation of spindling to bilateral SW activity in absence epilepsy. Recently Gelinas et al. using a rat kindling model, provided elegant experimental evidence for SPW-R’s conversion to epileptic variants as well as for IEDs’ interference with temporo-frontal memory consolidation. They have shown that the hippocampus displaying sleep related IEDs cannot process normal engrams [31,32].
In summary, the progressive transformation of SPW-Rs to spikes and fast ripples may be dynamic functional factors obstructing memory consolidation.
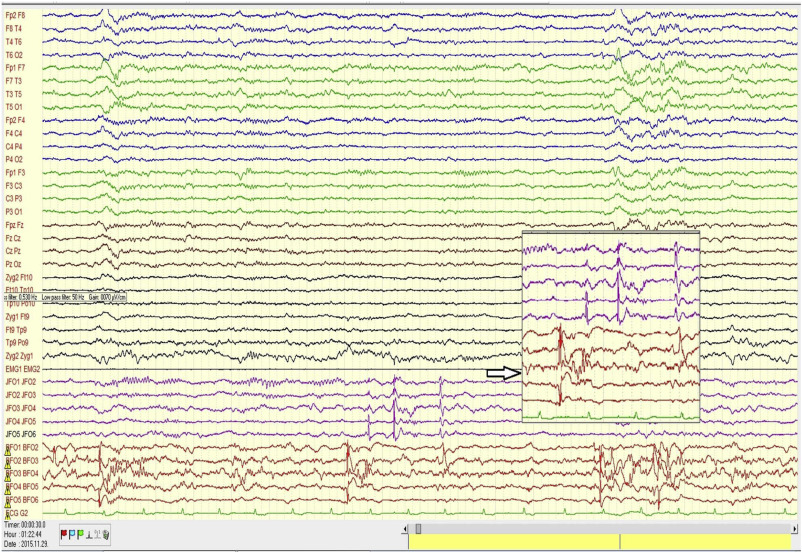
Figure 3. illustrates the abundant flow of discharges recorded in bilateral foramen ovale electrodes during sleep in one of our patients under pre-surgical evaluation.
Figure 3.
Scalp and bilateral foramen ovale (FO) records in a patient with bitemporal epilepsy. Note the intensive interictal spiking independently in both foramen ovale (FO) records. The flood of spikes in FO electrodes has no trace in the scalp electrodes. The spikes are conjoined several times by fast activity (not filtered for possible HFO, however, with more appropriate filtering, HFO could be made visible).
These results support that beyond structural hippocampal sclerosis, IEDs might contribute to the memory loss of MTLE patients by at least two mechanisms:
1) Daytime spikes obstruct the elaboration of normal engrams (a hypothesis).
2) Sleep-related epileptic discharges as dysfunctional dummies of SPW-Rs are unable to process and consolidate engrams [31,32].
Clinical data support that ictal and interictal epileptic activity cause memory loss.
Since the famous H.M case, undergoing bilateral hippocampectomy, the essential role of the paired hippocampi in human declarative memory has become obvious [33]. Due to the spread of surgical neuropsychology testing of TLE patients as well as to the progress of the neuro-imaging the learning of memory-production has improved. However, we do not see the entire mechanism and the whole harm caused by epilepsy. The scanty neuropsychological capacity mainly applied in surgical cases and, the lack of long-term follow-up studies limits our knowledge. Analyzing the structural damage of the hippocampus in line with hippocampal discharges would be especially useful.
Several ictal semiology phenomena reflect memory changes (e.g. déjà-vu and jamais-vu auras, pure amnestic fits etc.). One may wonder whether an abrupt drop of memory rather than a global disturbance of consciousness underlies patients’ loss of contact in their complex partial seizures.
The perisylvian cognitive network (PN) and idiopathic age-dependent focal/regional childhood epilepsies (IFCE). The malignant transformation to electrical status epilepticus in sleep (ESES) and Landau-Kleffner syndrome (LKS)
The PN is a large scale cognitive brain system of human communication e.g. speech, reading/writing and working memory, closely interconnected with the thalamus. Due to its delicate functions it is highly vulnerable to developmental errors.
IFCE as RE and PS are transient developmental conditions typically starting in schoolchildren and ending in puberty. This group constitutes the most prevalent type of epilepsy in childhood.
Rolandic epilepsy (age-dependent childhood epilepsy with centro-temporal spikes)
RE affects mentally normal children in the age group 2 to 13. The rare (1-2/year) seizures evolve during NREM sleep around sleep onset. Attacks strictly related to sleep occur in two third of patients. The original definition has excluded any lesional background; however, it may affect children with brain damages as well. In a study of non-lesional cases, neuroimaging revealed a thinner cortex in RE patients compared to controls in the frontal, temporal and, occipital regions [34], supporting a neurodevelopmental origin.
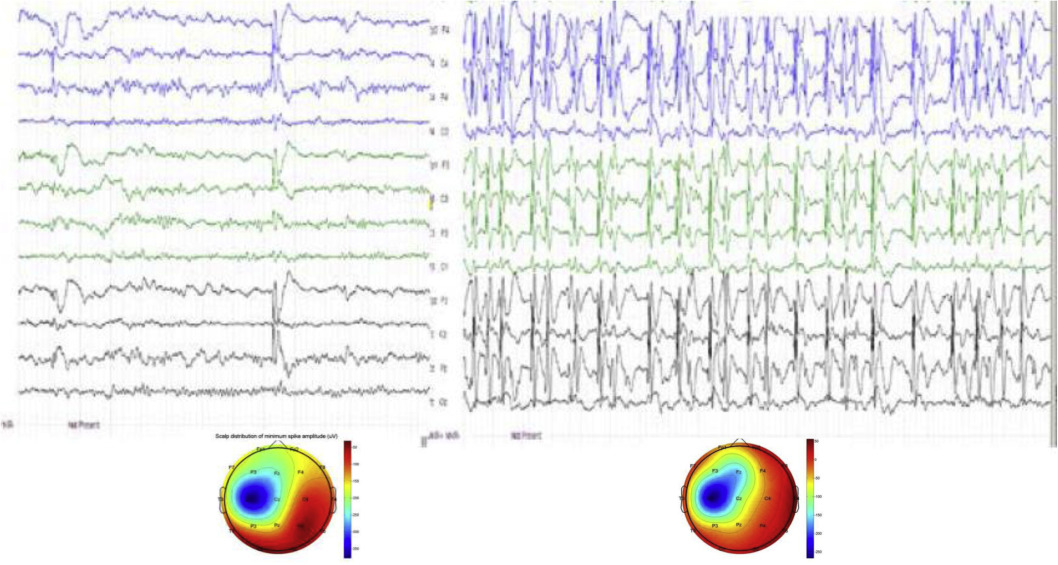
The EEG hallmark of the syndrome is the interictal CTS (Figure 4 a). It is a high voltage sharp wave closed by a slow wave in the mid-temporal region occurring in clusters or runs in a bilateral independent pattern in half of cases. The centrotemporal potential-field may shift to the frontal or parietal regions with unchanged seizure semiology. Typically, it is a fronto-temporal dipole, localized perpendicularly to the axis of the Sylvian fissure (Figure 4 b). Mapping studies in RE and PS patients [34-36] found the dipole of CTS around the big cortical sulci (Sylvian fissure, parieto-occipital sulcus and calcarine fissure in PS). CTSs are associated with 93.8-152.3 Hz ripples in RE [38] especially near seizures. In atypical cases turning to ESES, proportionately to the poor prognosis, the ripples had higher voltage.
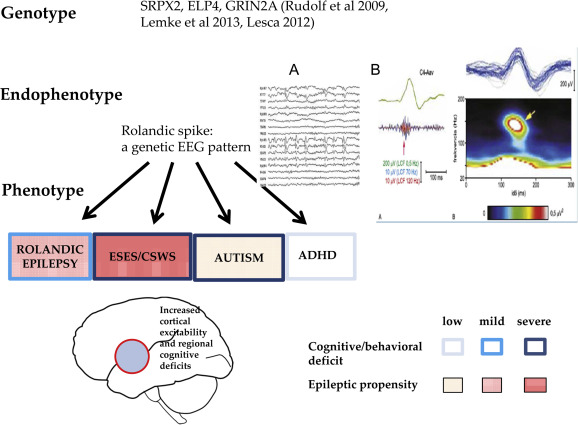
CTS as a shared sleep pattern in ICFE and ESES/LKS. TOP: the recognized genetic abnormalities. Middle: Endophynotype characteristics: A) CTS EEG pattern, B) CTS crowned by ripple (after Kobayashi 2011). Bottom: Phenotypic features. Schematic representation of epileptic propensity (red) and cognitive impairment (blue). The territory of the red circle represents the area of increased epileptic excitability and cognitive/behavioral deficits.
Slow wave sleep is the main enhancer of seizures and IEDs. Clemens and Majoros [75] found the strongest activation during deep slow wave sleep, especially on the descending slopes of the first sleep cycles. Beelke et al. and Nobili et al. [39,40] found a robust correlation of IEDs with spindling; unlike other groups of epilepsies where IEDs associate to the A1 phase of Cyclic Alternating Pattern (CAP).
About 1/4 of patients have one or more family members presenting with seizures and the non-epileptic first degree relatives of 1/3 of patients carry CTS. Also twin-studies have supported a genetic background. Recent studies highlighted the possible role of the SRPXA, GRIN2A and ELP4 genes [41-45], involved in cortical development.
Panayiotopoulos syndrome (PS)
Describing it in 1999, Panayiotopoulos further explicated the syndrome in an additional paper [46,47]. A 2006 consensus report characterized it as “benign, age-related, focal type of epilepsy developing in early and mid-childhood [48]. The interictal EEG consists of multiple/changing localization, most consequently occipital; morphologically BCTE-like discharges”. PS is prevalent in children younger than 6 constituting 13 % of all epilepsies. It may associate with RE.
In PS, mapping studies found potential fields around the occipito-medial and parieto-occipital regions as well as around the calcarine sulcus [36]. Seizures start in age 3 to 6 and cease around 14-15 years, occurring in sleep in 2/3 of cases [49]. The electro-morphologic features are similar to RE with variable localization and hemispheric distribution CTSs. NREM sleep has a less strong activating effect than in RE.
The seizures are not always stereotyped. Half of them last longer than 30 minutes and may aggregate to status epilepticus. Most seizures manifest with autonomic signs. Nausea, retch and vomiting are most frequent featuring most seizures. Color-changes, cyanosis, dilated pupils, temperature-swings, incontinence, salivation, vehement bowel movements and syncope may present as sole ictal phenomena or may be joined by convulsions or atony. Eye deviation as an initial symptom is less frequent than deemed earlier. The genetic background is unknown.
The transition to ESES/LKS
Since the seventies of the last century, several age-dependent childhood epilepsy patients with a malignant disease-turn appeared in the literature: the children became speech-loss or suffered from a global cognitive decline [48,49]. Their IEDs inundated the cortical convexities during all NREM sleep stages constituting electrical status epilepticus. This nearly continuous and widespread bilateral spiking during slow wave sleep may last for months or years without clinical seizures.
Tassinari et al called this malignant variant “encephalopathy related to electric status epilepticus” [50-52] now known as electrical status epilepticus in sleep (ESES). Its treatment has remained unresolved, the antiepileptic drugs are ineffective [53]. ESES may start in early childhood and cease before puberty [54,55]. The severity and reversibility of cognitive loss correlates with the duration and recovery of ESES . The degree and type of cognitive loss depend on the degree and localization of IEDs during NREM sleep . A deviant RE course may underlie ESES [53-58].
Acquired epileptic aphasia named Landau-Kleffner syndrome (LKS) honoring the first descriptors, is a regional variant of ESES, it is another malignant variant of PN conditions. The abundant spikes of LKS cluster in the posterior temporal region of the dominant hemisphere [59], with no underlying lesion. The affected children lose speech-perception after normal speech had evolved prior to the disorder [50,53,60].
Genetic findings support the notion of disease spectrum involving RE, PS, atypical RE, LKS and ESES [41,42].
We published an extensive conception called „The perisylvian epileptic network” on the spectrum of benign focal childhood epilepsies transforming to LKS or ESES [60]. In line with us, Fejerman [61] has analyzed the features of the malignant disease-course named by him „atypical Rolandic epilepsy”. Di Negri published a very similar unifying concept highlighting the spectrum-like nature of IFCE ranging from RE and PS to ESES and LKS [63].
The continuity of IEDs in perisylvian spectrum-disorders
The critical sleep-enhancement of CTS is a fundamental feature of the whole spectrum [64]. There is a continuity in the amount, synchronization, and bilateralisation of spikes [61] and fast ripples from the benign conditions (IFCE) to the malignant encephalopathic variants [38,89].
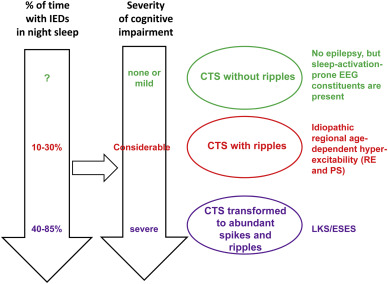
CTS have a stepwise evolution. If it presents without ripples, there is no clinical epilepsy. Ripples join CTS in the RE-PS phenotype (Figure 4. a, b) and in malignant cases developing to ESES/LKS abundant spikes and ripples occur (Figure 8.) [38].
Figure 5.
Transition from wake state (left) to NREM sleep (right) shows the appearance of abundant spiking (ESES) in an 8 year-old boy. Bottom: amplitude mapping: averages of 10 awake (left) and 100 sleep (right) spikes. Note the similar spatial and laterality patterns of the focal spike fields in wakefulness and sleeping.
Figure 6.
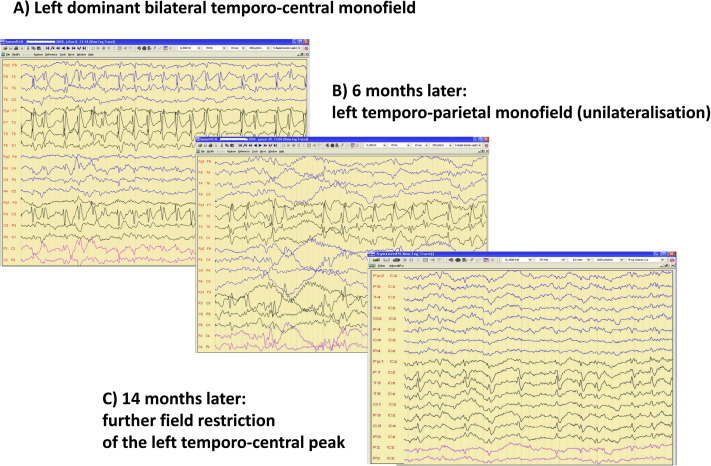
Regression of spiking during remission of ESES. A: Left dominant bilateral temporo-central monofield, B: 6 months later left temporo-parietal monofield (unilateralisation), C: 14 months later: more field restriction of the left temporo-central peak.
Figure 7.
The relationship between sleep oscillations and the occurrence rate of CTS among the spectrum of perisylvian epileptic network disorders. Top: Distribution of CTS across the sleep cycle. Bottom: Distribution of IEDs (green dashed line); the EEG sigma band (blue line = 12-16 Hz) and slow wave activity red line = 0.5-4.0 Hz) across the first NREM cycle. It is clearly seen that CTS goes parallel with sigma frequencies (sleep spindles), and not with sleep slow wave activity. (After Nobili et al (2011).
Figure 8.
Spectrum disorders of the perisylvian epileptic network. Continuity in the constituents across disorders (syndromes) is shown. Right: the centro-remporal spike (CTS) which is the basic endophenotype of the spectrum. The three levels of CTS characteristics in clinical syndromes: green= CTS without ripples corresponds to lack of epileptic seizures; red=CTS with ripples corresponds to idiopathic regional age dependent hyper-excitability (Rolandic epilepsy and Panayitopoulos syndrome); purple = abundance of CTS-like discharges in sleep with ripples corresponds to ESES/LKS. Left: The perpendicular arrows represent the increasing amount of epileptic activity in sleep and increasing degree of cognitive impairment. The horizontal black arrow represents the assumed causal relationship between sleep epileptic activity and cognitive impairment. IEDs: interictal epileptic discharges.
Endophenotypes are shared modules of phenotypically different complex disorders e.g. in schizophrenia and in certain epilepsies. CTS is an endophenotype shared by RE, PS, ESES/LKS. It is more prevalent in the sleep of autism spectrum disorder [63,64] and ADHD [66,67] patients than in the normal population (Figure 4.). Tittering upon the edge of non-epilepsy and epilepsy, it has a vague nature. Non-epileptic CTS present without ripples in autism [64,65] and ADHD [66,67]. It behaves as an augmented evoked potential responding to acoustic and tactile stimuli [68,69] in certain infants, while epileptic CTS in RE are joined by fast ripples HFO [38,63,70].
Recently Mirandola et al. [71] published a case report on a thirteen year-old boy presenting with moderate learning difficulties affecting reading, writing and calculation. He had no clinical epilepsy but right dominant bilateral independent CTS with important enhancement during NREM sleep, not reaching the 85% cut-off for ESES. FMRI In wakefulness revealed a right dominant increment of the BOLD signal in the sensory-motor cortex, while during NREM sleep, it discovered a widespread CTS-related cortical-subcortical network over the thalamic-perisylvian region. This case shows that CTS during NREM sleep may link with a large pathological network with no epilepsy and it carries a risk of cognitive decline.
Thus, CTS might be the EEG sign of local cortical developmental delay and increased local excitability [46,72,73] with a potential to regress or progress to epilepsy and its malignant variants.
The continuity of sleep enhancement in PN spectrum disorders
In cases where RE and PS patients had had sleep recordings prior to their progression to ESES; their IEDs were similar to those later seen in ESES; just quantitative differences occurred. This supports a continuity in the degree of sleep activation as well. In addition, the abundant discharges of ESES in sleep versus CTS in waking constitute similar potential-fields (Figure 5.). While the activation rate of CTS without ripples during sleep is unknown, CTS with ripples strongly activate during sleep in RE patients [72-74]. The earlier rigorous 85% covering of NREM sleep by IEDs as the criterion for diagnosing ESES has become more flexible recently; an important continuous regional, hemispheric or global activation of IEDS is accepted. Thus, the view on ESES as a unique entity has turned to a spectral range concept. There are continual features in time and territory of sleep activation, during the evolution and remission of ESES, too [75] (Figure 6.).
There is another important feature differentiating PN spectrum conditions (IFCE) from the rest of sleep-related epilepsies. In PN conditions, CTS couple with sleep spindling [76,77,39,40] (Figure 7.) unlike in other epilepsies where spikes link with CAP A1 phase slow waves [78].
The continuity of cognitive impairment in PN conditions
In contrast with the early expectation on a benign outcome of IFCE, several recent publications revealed a wide range of associated cognitive and behavioral disturbances. A systematic review and meta-analysis of 42 case-control samples comprising 1237 untreated RE children and 1137 healthy controls showed significantly lower scores in eight cognitive domains, most importantly in long term storage and retrieval [79]. Other studies found cognitive impairment phenotypes affecting language and academic performance [80]. Bilateral discharges associated to more severe cognitive impairment [81,82].
The cognitive changes found in RE and PS fill the gap between them and their atypical variants progressing to ESES and LKS. A recent conference on idiopathic focal epilepsies “The lost tribe state of the art” [83] supports this approach in a multilateral way.
Shared genetic background
PN spectrum conditions share several genetic features. Recent research found the SRPX2 and ELP4 genes involved in epileptic sleep activation, possibly through influencing cell motility, migration and, adhesion [41]. Lemke et al [45] revealed changes in the gene encoding the NMDA receptor NR2A subunit as a major genetic risk factor of PN spectrum-conditions. They found new heterozygous mutations in GRIN2A in 27 out of 359 affected individuals from two independent cohorts with growing frequency toward the ESES end.
In the RE-ESES/LKS spectrum, increased copy-number variations were found in the genomic architecture of several genes encoding cell adhesion proteins and there are some hints pointing the autism spectrum as well [42].
The continuity of PN spectrum conditions and the evolution of the syndrome constituents is summarized in Figure 8.
Other infantile and childhood encephalopathies
Epileptic encephalopathies (EEs) (early infantile epileptic encephalopathies, West-syndrome, Lennox-Gastaut syndrome) constitute a contradictory however, widely used group in epileptology; recognized by the International League against Epilepsy. EEs are epilepsy-related conditions of early-affected large brain systems, likely involving the cortico-thalamic system. The main criterion is that the associated cognitive symptoms are due to epilepsy, encephalopathies caused by brain lesions and drugs are not included in this group. There is a conceptual difference between EEs and encephalopathies with epilepsy. In the latter group, a common etiology e.g. tuberous sclerosis underlies epilepsy and cognitive symptoms, while for example in ESES, sleep-related IEDs cause a cognitive loss.
The concept of system epilepsies overlaps with EEs [84,85]. For understanding the two steps in the evolution of EEs, the term “developmental epileptic encephalopathies” was proposed recently [86].
The weakness of the EEs category is that it might blur the actual mechanism of cognitive decline [87,88].
EEs typically start in infancy or early childhood. The specific combination of symptoms constituting different EEs might have a common pathomechanism caused by variable etiologies. Some EEs may have a genetic background. There is a progressive evolution to the full-blown picture in most cases; becoming stagnant and resistant to treatment after. In a sensitive early age, the epileptic disorder takes over the organization of physiological networks involved in plasticity e.g. in Lennox-Gastaut syndrome [89].
Childhood EEs share abundant IEDs during NREM sleep. The suppression burst activity; a sleep-variant of hypsarrhythmia covering the whole sleep in early neonatal encephalopathies likely interferes with normal brain development. It disfigures sleep in West syndrome. The frequent and variable topography seizures contribute to the cognitive harm caused by IEDs. The cognitive improvement experienced when Vigabatrine helps hypsarrhythmia and epileptic spasms, supports their causative role in cognitive loss. The diffuse slow SWs and generalized fast paroxysmal activity with or without tonic seizures in Lennox-Gastaut syndrome interfere with normal cognitive functioning as well.
Even scalp electrodes can detect now the abundant fast ripples during NREM sleep [90]. There are fast ripples in most childhood EEs, correlating with the severity and progression of EE [38,91] and offering another hint of their impact on cognitive impairment. Because the cut-off frequencies of fast ripples in infants and children are not yet clear, conclusions regarding the significance of different frequency-ranges should be taken cautiously.
In MTLE and PN spectrum conditions there are relevant studies supporting the interference of epileptic transformation with plastic sleep functions and cognition. There are much less data in EEs, but one may suspect similar mechanisms underlying the cognitive decline in EE patients.
We summarize the common features of childhood encephalopathies in Table 1.
Table 1
The common features of epileptic encephalopathies.
|
• Most of them are age-related, starting in early childhood, in the period of high brain plasticity. |
|
• Cognitive dysfunction is supposed to be underlain by the disordered association cortex. |
|
• There is pathological HFO (fast ripples) during sleep in all forms. |
|
• Interictal epileptic activity has a more important harm than seizures themselves. |
|
• Epileptic encephalopathies constitute a spectrum of an age dependent syndrome chain (Ohtahara syndrome-West syndrome- Lennox-Gastaut syndrome). |
|
• Because several epileptic encephalopathy patients have genetic, metabolic, hypoxic or other brain damages, it is hard to identify those cognitive deficits caused by epilepsy itself. |
Discussion
In this paper, we review three types of epilepsies disfiguring major brain systems by the derailment of NREM sleep circuits.
The first one is temporal lobe epilepsy where SPW-Rs involved in memory processing transform to spikes and pathological HFO (fast ripples) compromising the declarative memory system.
The second example is the group of IFCE progressing to ESES/LKS characterized by severe cognitive decline.
The third one is the group of EEs, where the abundant spiking and HFO (mainly inNREM sleep) interfere with sleep plastic functions.
In our recent book [62] we have reviewed these transformations more comprehensively. We suggested a common way how certain major epilepsies compromise sleep plastic functions causing cognitive impairment. This key mechanism is the consequence of the plasticity-dependent strong bond between NREM sleep and epilepsy and provides an angle for learning developmental epilepsies.
We are on the way to see why the cognitive decline in epilepsies associates mainly with IEDs of NREM sleep [9-13] in accordance with the synaptic homeostasis theory [92].
Sleep-related memory consolidation makes slow wave sleep the pledge of learning, justifying the need and seemingly uneconomical presence and persistence of sleep across the phylogeny. IEDs interweaving NREM sleep may interfere with its plastic functions; vividly depicted by Tassinari in the description of „Penelope syndrome”* [93].
A vicious circle can be delineated in the sleep-epilepsy-cognition triangle: slow wave sleep is distorted by IEDs compromising normal sleep functions. Thus the cognitive harm is not the direct consequence of epilepsy, rather its indirect complication through the interference with sleep plasticity. This important cascade of events is increasingly recognized providing an explanation of cognitive decline in EEs, in addition to lesional and pharmacological factors.
Recent studies on the sleep of ESES patients indirectly suggest the deficit of night-time synaptic down-scaling [9,10,12,92]. Recently Bölsterli et al [10] have shown that after the recovery from ESES the night-time decline of slow waves restored, and the outcome was better if sleep slow wave decline had remained somewhat preserved during the active ESES period.
There seem to be two different ways of memory-harm caused by IEDs. In the one, spikes interfere with memory consolidation during NREM sleep e.g. in MTLE. In the second one, IEDs interfere with the general synaptic down-scaling process during NREM sleep e.g. in ESES and LKS.
Recent research links epilepsy to network activities; considering interictal and ictal signs as network states. Ictal states represent pathological peaks of activity, expressed by episodic changes of behavior; interictal states constitute a more continuous background of the disorder.
* In the legendary epos of Homer “The Trojan war” during the absence of Odysseus, Penelope his wife told to her proposers that she needed to weave her father-in-law’s Laertes funeral shroud first, she could choose from among them only after. What she weaved during the day, she unraveled secretly during each night, delaying the time of decision (Analogy: what is built up in the neuronal networks during the day cannot be utilized due to an interference by night-time spiking).
The influence of this hidden continuous and insidious activity manifesting no spectacular events has remained more or less unknown even after many years of intensive epilepsy research. We certainly need a switch from dealing with transient to long-term (and possibly progressive) cognitive impairment. Performing repeated sleep studies combined with cognitive follow up studies is hard; requiring highly organized cooperative multi-center work embracing decades, and this is before us.
In this paper we used the system-epilepsy approach to demonstrate how sleep plastic functions are compromised by the epileptic derailment occupying („hijacking” [94]) them. The concept of system-epilepsy [84,85] comes from the now generally accepted notion of epilepsy as a neuronal network disease. Most neural networks serve certain physiological systems. Major epilepsies can be conceived as physiological systems in which an epileptic over-excitation (derailment) exaggerates the functions when they are activated. Sensory or praxis induced reflex epilepsies may be reinterpreted so [Szücs et al submitted], and certain non-sensory systems like sleep, arousal or the memory system might be additional examples [95].
One of the advantages of the system epilepsy approach is, that it may highlight the interictal symptoms of epilepsy as memory disturbances and other cognitive deficits. The ictal signs are more emphasized nowadays (mainly due to the highly developing epilepsy surgery) while interictal ones remain in shadow compared to their significance.
Conflict of interest
The authors have no conflict of interest to declare.
Practice Points
- Due to the link of epilepsy and NREM sleep, sleep EEG is indispensable in the evaluation of epilepsies.
- Sleep-relatedness and interictal epileptic activity have major role in chronic cognitive harm.
- Sleep EEG may clarify the cause of unexplained childhood cognitive decline or dysphasia.
Research Agenda
- Assessing the share of spiking and structural hippocampal damage in the cognitive loss of MTLE.
- Cognitive impairment needs to be correlated with the enhancement of sleep spiking in conditions with cognitive decline.
- There is pressing need to find pharmaco-therapy treatment against interictal epileptic activity.
Acknowledgement
The authors are grateful for the support of the National Brain Research Program 2017-1.2.1-NKP-2017-00002.gs1
References
1). Zhao F., Kang H., You L., Rastogi P., Venkatesh D., and Chandra M.: Neuropsychological deficits in temporal lobe epilepsy: A comprehensive review. Ann Indian Acad Neurol 2014; 17: pp. 374-382
2). Holmes G.L.: What is more harmful, seizures or epileptic EEG abnormalities? Is there any clinical data? Epileptic Disord 2014; 1: pp. S12-S22
3). Binnie C.D.: Significance and management of transitory cognitive impairment due to subclinical EEG discharges in children. Brain Dev 1993; 15: pp. 23-30
4). Binnie C.D.: Cognitive effects of subclinical EEG discharges. Neurophysiol Cli 1996; 26: pp. 138-142
5). Halász P.: The relationship of medial temporal lobe epilepsy with the declarative memory system. J Epileptology 2016; 24: pp. 157-165
6). Shatskikh T.N., Rag havendra M., Zhao Q., Cui Z., and Holmes G.L.: Electrical induction of spikes in the hippocampus impairs recognition capacity and spatial memory in rats. Epilepsy Behav 2006; 9: pp. 549-556
7). Clemens Z., Mölle M., Erőss L., Barsi P., Halász P., and Born J.: Temporal coupling of parahippocampal ripples, sleep spindles and slow oscillations in humans. Brain 2007; 130: pp. 2868-2878
8). Kleen J.K., Scott R.C., Holmes G.L., Roberts D.W., Rundle M.M., Testorf M., et al: Hippocampal interictal epileptiform activity disrupts cognition in humans. Neurology 2013; 81: pp. 18-24
9). Bölsterli B.K., Schmitt B., Bast T., Critelli H., Heinzle J., Jenni O.G., and Huber R.: Impaired slow wave sleep downscaling in encephalopathy with status epilepticus during sleep (ESES). Clin Neurophysiol 2011; 9: pp. 1779-1787
10). Bölsterli B.K., Gardella E., Pavlidis E., Wehrle F.M., Tassinari C.A., Huber R., et al: Remission of encephalopathy with status epilepticus (ESES) during sleep renormalizes regulation of slow wave sleep. Epilepsia 2017; 58: pp. 1892-1901
11). Bölsterli B.K., Fattinger S., Kurth S., LeBourgeois M.K., Ringli M., Bast T., et al: Spike wave location and density disturb sleep slow waves in patients with CSWS (continuous spike waves during sleep). Epilepsia 2014; 55: pp. 584-591
12). Cantalupo G., Rubboli G., and Tassinari C.A.: Night-time unravelling of the brain web: impaired synaptic downscaling in ESES-the Penelope syndrome. Clin Neurophysiol 2011; 122: pp. 1691-1692
13). Rasch B., and Born J.: About Sleep’s Role in Memory. Physiol Rev 2013; 93: pp. 681-766
14). García-Peñas J.J.: Interictal epileptiform discharges and cognitive impairment in children. Rev Neurol 2011; 1: pp. S43-S52
15). Buzsáki G.: Hippocampal Sharp Wave-Ripple: A cognitive Biomarker for Episodic Memory and Planning. Hippocampus 2015; 25: pp. 1073-1188
16). Buzsáki G.: Hippocampal sharp waves their origin and significance. Brain Res 1986; 398: pp. 242-252
17). Buzsáki G.: Two-stage model of memory trace formation: a role for “noisy” brain states. Neuroscience 1989; 31: pp. 551-570
18). Chrobak J.J., and Buzsáki G.: Operational dynamics in the hippocampal-entorhinal axis. Neurosci Biobehav Rev 1998; 22: pp. 303-310
19). Csicsvári J., Hirase H., Czurkó A., Mamiya A., and Buzsáki G.: Oscillatory coupling of hippocampal pyramidal cells and interneurons in the behaving rat. J Neurosci 1999; 19: pp. 274-287
20). Schlingloff D., Káli S., Freund T.F., Hájos N., and Gulyás A.I.: Mechanisms of sharp wave initiation and ripple generation. J Neurosci 2014; 20: pp. 11385-11398
21). Gulyás A.I., and Freund T.T.: Generation of physiological and pathological high frequency oscillations: the role of perisomatic inhibition in sharp-wave ripple and interictal spike generation. Curr Opin Neurobiol 2015; 31: pp. 26-32
22). Girardeau G., Benchenane K., Wiener S.I., Buzsáki G., and Zugaro M.B.: Selective suppression of hippocampal ripples impairs spatial memory. Nat Neurosci 2009; 12: pp. 1222-1223
23). Roux L., Hu B., Eichler R., Stark E., and Buzsáki G.: Sharp wave ripples during learning stabilize the hippocampal map. Nat Neurosci 2017; 20: pp. 845-853
24). Nádasdy Z.: Spike sequences and their consequences. J Physiol Paris 2000; 94: pp. 505-524
25). Foster D.J., and Wilson M.A.: Reverse replay of behavioural sequences in hippocampal place cells during the awake state. Nature 2006; 440: pp. 680-683
26). Ji D., and Wilson M.A.: Coordinated memory replay in the visual cortex and hippocampus during sleep. Nat Neurosci 2007; 10: pp. 100-107
27). Cheng S., and Frank L.M.: New experiences enhance coordinated neural activity in the hippocampus. Neuron 2008; 7: pp. 303-313
28). O’Neil J., Senior T.J., Allen K., Huxter J.R., and Csicsvári J.: Reactivation of experience-dependent cell assembly patterns in the hippocampus. Nat Neurosci 2008; 11: pp. 209-215
29). Frauscher B., von Ellenrieder N., Ferrari-Marinho T., Avoli M., Dubeau F., and Gotman J.: Facilitation of epileptic activity during sleep is mediated by high amplitude slow waves. Brain 2015; 138: pp. 1629-1641
30). Frauscher B., Bernasconi N., Caldairou B., von Ellenrieder N., Bernasconi A., Gotman J., and Dubeau F.: Interictal Hippocampal Spiking Influences the Occurrence of Hippocampal Sleep Spindles. Sleep 2015; 38: pp. 1927-1933
31). Gelinas J.N., Khodagholy D., Thesen T., Devinsky O., and Buzsáki G.: Interictal epileptiform discharges induce hippocampal-cortical coupling in temporal lobe epilepsy. Nat Med 2016; 22: pp. 641-648
32). Boly M., Jones B., Findlay G., Plumley E., Mensen A., Hermann B., Tononi G., and Maganti R.: Altered sleep homeostasis correlates with cognitive impairment in patients with focal epilepsy. Brain 2017; 140: pp. 1026-1040
33). Milner B.: The medial temporal-lobe amnesic syndrome. Psychiatr Clin North Am 2005; 28: pp. 599-611
34). Vannest J., Tenney J.R., Gelineau-Morel R., Maloney T., and Glauser T.A.: Cognitive and behavioral outcomes in benign childhood epilepsy with centrotemporal spikes. Epilepsy Behav 2015; 45: pp. 85-91
35). Wong P.K.: Source modelling of the Rolandic focus. Brain Topogr 1991; 4: pp. 105-112
36). Kanazawa O., Tohyama J., Akasaka N., and Kamimura T.: A magnetoencephalographic study of patients with Panayiotopoulos syndrome. Epilepsia 2005; 46: pp. 1106-1113
37). Yoshinaga H., Koutroumanidis M., Kobayashi K., Shirasawa A., Kikumoto K., Inoue T., Oka M., and Ohtsuka Y.: EEG dipole characteristics in Panayiotopoulos syndrome. Epilepsia 2006; 47: pp. 781-787
38). Kobayashi K., Yoshinaga H., Toda Y., Inoue T., and Oka M.: High-frequency oscillations in idiopathic partial epilepsy of childhood. Epilepsia 2011; 52: pp. 1812-1819
39). Beelke M., Nobili L., Baglietto M.G., De Carli F., Robert A., et al: Relationship of sigma activity to sleep interictal epileptic discharges: a study in children affected by benign epilepsy with occipital paroxysms. Epilepsy Res 2000; 40: pp. 179-186
40). Nobili L., Baglietto M.G., Beelke M., De Carli F., De Negri E., Gaggero R., et al: Distribution of epileptiform discharges during nREM sleep in the CSWSS syndrome: relationship with sigma and delta activities. Epilepsy Res 2001; 44: pp. 119-128
41). Rudolf G., Valenti M.P., Hirsch E., and Szepetowski P.: From rolandic epilepsy to continuous spike- and-waves during sleep and Landau-Kleffner syndromes: insights into possible genetic actors. Epilepsia 2009; 50: pp. 25-28
42). Lesca G., Rudolf G., Bruneau N., Lozovaya N., Labalme A., Boutry-Kryza N., et al: GRIN2A mutations in acquired epileptic aphasia and related childhood focal epilepsies and encephalopathies with speech and language dysfunction. Nat Genet 2013; 45: pp. 1061-1066
43). Reutlinger C., Helbig I., Gawelczyk B., Subero J.I., Tönnies H., et al: Deletions in 16p13 including GRIN2A in pat ients with intellectual disability various dysmorphic features, and seizure disorders of the rolandic region. Epilepsia 2010; 1: pp. 1870-1873
44). Dimassi S., Labalme A., Lesca G., Rudolf G., Bruneau N., et al: A subset of genomic alterations detected in Rolandic epilepsies contains candidate or known epilepsy genes including GRIN2A and PRRT2. Epilepsia 2014; 55: pp. 370-378
45). Lemke J.R., Lal D., Reinthaler E.M., Steiner I., Nothnagel M., Alber M., et al: Mutations in GRIN2A cause idiopathic focal epilepsy with Rolandic spikes. Nat Genet 2013; 45: pp. 1067-1072
46). Panayiotopoulos C.P.: Early-onset benign childhood occipital seizure susceptibility syndrome: a syndrome to recognize. Epilepsia 1999; 40: pp. 621-630
47). Panayiotopoulos CP ed. Panayiotopoulos syndrome: a common and benign childhood epileptic syndrome. London: John Libbey, Eastleigh, 2002.
48). Ferrie C., Caraballo R., Covanis A., Demirbilek V., Dervent A., Kivity S., et al: Panayiotopoulos syndrome: a consensus view. Dev Med Child Neurol 2006; 48: pp. 236-240
49). Caraballo R., Cersósimo R., and Fejerman N.: Panayiotopoulos syndrome: a prospective study of 192 patients. Epilepsia 2007; 48: pp. 1054-1061
50). Patry G., Lyagoubi S., and Tassinari C.A.: Subclinical electrical status epilepticus induced by sleep in children. Arch Neurol 1971; 24: pp. 242-252
51). Tassinari C.A., Rubboli G., Volpi L., Meletti S., d’Orsi G., Franca M., Sabetta A.R., Riguzzi P., Gardella E., Zaniboni A., and Michelucci R.: Encephalopathy with electrical status epilepticus during slow sleep or ESES syndrome including the acquired aphasia. Clin Neurophysiol 2000; 111: pp. S94-S102
52). Tassinari C.A., Dravet C., and Roger J.: Encephalopathy related to electrical status epilepticus during slow sleep. Electroenceph Clin Neurophysiol 1977; 43: pp. 529-530
53). Kramer U., Sagi L., Goldberg-Stern H., Zelnik N., Nissenkorn A., and Ben-Zeev B.: Clinical spectrum and medical treatment of children with electrical status epilepticus in sleep (ESES). Epilepsia 2009; 50: pp. 1517-1524
54). Sanchez Fernandez I., Peters J.M., Hadjiloizou S., Prabhu S.P., Zarowski M., Stannard K.M., Takeoka M., Rotenberg A., Kothare S.V., and Loddenkemper T.: Clinical staging and electroencephalographic evolution of continuous spikes and waves during sleep. Epilepsia 2012; 53: pp. 1185-1195
55). Sánchez Fernández I., Loddenkemper T., Peters J.M., and Kothare S.V.: Electrical status epilepticus in sleep: clinical presentation and pathophysiology. Pediatr Neurol 2012; 47: pp. 390-410
56). Dalla Bernardina B., Tassinari C.A., Dravet C., Bureau M., Beghini G., and Roger J.: Benign focal epilepsy and “electrical status epilepticus” during sleep. Rev Electroencephalogr. Neurophysiol Clin 1978; 8: pp. 350-353
57). Dalla Bernardina B., Sgrò V., Caraballo R., Fontana E., Colamaria V., Zullini E., et al: Sleep and benign partial epilepsies of childhood: EEG and evoked potentials study. Epilepsy Res Suppl 1991; 2: pp. 83-96
58). Saltik S., Uluduz D., Cokar O., Demirbilek V., and Dervent A.: A clinical and EEG study on idiopathic partial epilepsies with evolution into ESES spectrum disorders. Epilepsia 2005; 46: pp. 524-533
59). Kellermann K.: Recurrent aphasia with subclinical bioelectric status epilepticus during sleep. Eur J Pediatr 1978; 128: pp. 207-212
60). Halász P., Kelemen A., Clemens B., Saracz J., Rosdy B., Rásonyi G., and Szücs A.: The perisylvian epileptic network. A unifying concept. Ideggyogy Sz 2005; 58: pp. 21-31
61). Fejerman N.: Atypical rolandic epilepsy. Epilepsia 2009; 50: pp. 9-12
62). Halász P, Szűcs A. Sleep, Epilepsies, and Cognitive Impairment. Academic Press Elsevier, 2018. *
63). Di Negri : Electrical status epilepticus during sleep (ESES). Different clinical syndromes: towards a unifying view? Brain Dev 1997; 19: pp. 447-451
64). Lee Y.J., Hwang S.K., and Kwon S.: The Clinical Spectrum of Benign Epilepsy with Centro-Temporal Spikes: a Challenge in Categorization and Predictability. J 2017; 7: pp. 1-6
65). Rossi P.G., Parmeggiani A., Bach V., Santucci M., and Visconti P.: EEG features and epilepsy in patients with autism. Brain Dev 1995; 17: pp. 169-174
66). Nass R., and Devinsky O.: Autistic regression with rolandic spikes. Neuropsychiatry 1999; 12: pp. 193-197
67). Silvestri R., Gagliano A., Calarese T., Aricò I., Cedro C., Condurso R., et al: Ictal and interictal EEG abnormalities in ADHD children recorded over night by video-polysomnography. Epilepsy Res 2007; 75: pp. 130-137
68). Danhofer P., Pejčochová J., Dušek L., Rektor I., and Ošlejšková H.: The influence of EEG-detected nocturnal centrotemporal discharges on the expression of core symptoms of ADHD in children with benign childhood epilepsy with centrotemporal spikes (BCECTS): A prospective study in a tertiary referral center. Epilepsy Behav 2018; 79: pp. 75-81
69). Tassinari C.A., De Marco P., Plasmati R., Pantieri R., Bianco M., and Michelucci R.: Extreme somatosensory evoked potentials (ESEPs) elicited by tapping of hands or feet in children: a somatosensory cerebral evoked potentials study. Neurophysiol Clin 1988; 18: pp. 123-128
70). Fonesca L.C., and Tedrus G.M.: Epileptic syndromes in children with somatosensory evoked spikes. Clin. Electroencepalogr 1994; 25: pp. 54-58
71). Klink N.E.C., van‘t Klooster M.A., Leijten F.S.S., Jacobs J., Braun K.P.J., and Zijlmans M.: Ripples on rolandic spikes: A marker of epilepsy severity. Epilepsia 2016; 57: pp. 1179-1189
72). Mirandola L., Cantalupo G., Vaudano A.E., Avanzini P., Ruggier A., Pisani F., et al: Centrotemporal spikes during NREM sleep: The promoting action of thalamus revealed by simultaneous EEG and fMRI coregistration. Epilepsy Behav Case Rep 2013; 1: pp. 106-109
73). Doose H., and Baier W.K.: Benign partial epilepsy and related conditions: multifactoral pathogenesis with hereditary impairment of brain maturation. Eur J Pediatr 1989; 149: pp. 152-158
74). Koutroumanidis M., and Panayiotopoulos C.P.: Benign childhood partial epilepsies: benign childhood seizure susceptibility syndromes. J Neurol Neurosurg Psychiat 1993; 56: pp. 2-5
75). Clemens B., and Majoros E.: Sleep studies in benign epilepsy of childhood with rolandic spikes. II. Analysis of discharge frequency and its relation to sleep dynamics. Epilepsia 1987; 28: pp. 24-27
76). Halász P., Hegyi M., Siegler Zs , and Fogarasi A.: Encephalopathy with Electrical Status Epilepticus in Slow Wave Sleep – a review with an emphasis on regional (perisylvian) aspects. J Epileptology 2014; 22: pp. 71-87
77). Nobili L., Ferrillo F., Baglietto M.G., Beelke M., De Carli F., De Negri E., et al: Relationship of sleep interictal epileptiform discharges to sigma activity (12-16 Hz) in benign epilepsy of childhood with rolandic spikes. Clin Neurophysiol 1999; 110: pp. 39-46
78). Kellaway P.: The electroencephalographic features of benign centrotemporal (rolandic) epilepsy of childhood. Epilepsia 2000; 41: pp. 1053-1056
79). Terzano M.G., Parrino L., Spaggiari M.C., Barusi R., and Simeoni S.: Discriminatory effect of cyclic alternating pattern in focal lesional and benign rolandic interictal spikes during sleep. Epilepsia 1991; 32: pp. 616-628
80). Wickens S., Bowden S.C., and D’Souza W.: Cognitive functioning in children with self-limited epilepsy with centrotemporal spikes: A systematic review and meta-analysis. Epilepsia 2017; 58: pp. 1673-1685
81). Neumann H., Helmke F., Thiels C., Polster T., Selzer L.M., Daseking M., et al: Development in Children with Benign Rolandic Epilepsy of Childhood with Centrotemporal Spikes – Results of a Current Systematic Database Search. Fortschr Neurol Psychiatr 2016; 84: pp. 617-632
82). Chan S., Pressler R., Boyd S.G., Baldeweg T., and Cross J.H.: Does sleep benefit memory consolidation in children with focal epilepsy? Epilepsia 2017; 58: pp. 456-466
83). Pal D.K., Ferrie C., Addis L., Akiyama T., Capovilla G., Caraballo R., et al: Idiopathic focal epilepsies: the “lost tribe. Epileptic Disord 2016; 18: pp. 252-288
84). Avanzini G., Manganotti P., Meletti S., Moshé S.L., Panzica F., Wolf P., et al: The system epilepsies: a pathophysiological hypothesis. Epilepsia 2012; 53: pp. 771-778
85). Capovilla G., Moshé S.L., Wolf P., and Avanzini G.: Epileptic encephalopathy as models of system epilepsy. Epilepsia 2013; 54: pp. 34-37
86). Scheffer I.E., Berkovic S., Capovilla G., Connolly M.B., French J., Guilhoto L., et al: ILAE classification of the epilepsies: Position paper of the ILAE Commission for Classification and Terminology. Epilepsia 2017; 58: pp. 512-521
87). Howell K.B., Harvey A.S., and Archer J.S.: Epileptic encephalopathy: Use and misuse of a clinical syndrome: considerations and hypotheses. Epileptic Disord 2001; 3: pp. 183-196
88). Jehi L., Wyll ie E., and Devinsky O.: Epileptic encephalopathies: Optimizing seizure control and developmental outcome. Epilepsi 2015; 56: pp. 1486-1489
89). Blume W.T.: Pathogenesis of Lennox-Gastaut syndrome: considerations and hypotheses. Epileptic Disord 2001; 3: pp. 183
90). Frauscher B., Bartolomei F., Kobayashi K., Cimbalnik J., van ‘t Klooster M.A., Rampp S., et al: High-frequency oscillations: The state of clinical research. Epilepsia 2017; 58: pp. 1316-1329
91). Endoh F., Yoshinaga H., Ishizaki Y., Oka M., Kobayashi K., and Ohtsuka Y.: Abnormal fast activity before the onset of West syndrome. Neuropediatrics 2011; 42: pp. 51-54
92). Tononi G., and Cirelli C.: Sleep and synaptic homeostasis: a hypothesis. Brain Res Bull 2003; 62: pp. 143-150
93). Tassinari C.A., Cantalupo G., Rios-Pohl L., Giustina E.D., and Rubboli G.: Encephalopathy with status epilepticus during slow sleep: “the Penelope syndrome. Epilepsia 2009; 50: pp. 4-8
94). Beenhakker M.R., and Huguenard J.R.: Neurones that fire together also conspire together; is normal sleep circuitry hijacked to genrate epilepsy? Neuron 2009 11; 62: pp. 612-632
95). Halász P.: Are Absence epilepsy and Nocturnal Frontal Lobe Epilepsy System Epilepsies of the Sleep/Wake System? Behav. Neurol. 2015; undefined: pp. 231676
Sleep Medicine Reviews, Copyright © 2019