|
National Institute of Psychiatry and Neurology Epilepsy Centre, National Stroke Centre, Budapest, Hungary Correspondence: Anna Szűcs Received: February 15, 2001 Accepted: July 25, 2001 Key Words:Obstructive sleep apnoea, Haemorrhagic stroke, Ischaemic stroke follow-up, MESAM AbstractFrequency and severity of sleep apnoeas, snoring history, standard clinical stroke scale were assessed in 106 acute (73 ischaemic, 33 haemorrhagic) stroke patients. Thirty-seven patients with ischaemic stroke and 14 patients with cerebral bleeding, each having pathological oxygen desaturation index during sleep, were re-tested in 3 months for sleep apnoeas and clinical stroke scale. In haemorrhagic strokes, the apnoea frequency decreased parallel with clinical improvement; but it remained as frequent as in the acute phase in the ischaemic stroke group (p = 0.0002). Apnoea frequency decreased mostly during the course of posterior stroke (p = 0.0001). It is concluded that pathological sleep apnoea frequency remains stable after ischaemic stroke indicating a concomitant obstructive sleep apnoea syndrome and sleep apnoea is a transitory symptom of haemorrhagic strokes. IntroductionObstructive sleep apnoea syndrome (OSAS) is frequent in the elderly (5–40%) [1–3]. A strong association between OSAS and stroke morbidity is documented [4–11]. Depending on different OSAS criteria, it could be as frequent as 32–71% in acute stroke [12–16]. It is reported that sleep apnoea patients have a less favourable functional outcome of stroke and a higher mortality rate [13]. It is still an open question if OSAS is the cause or consequence [17–19] of cerebrovascular disorders. During the course of OSAS, sequential pathophysiological events occur, which can compromise the brain circulation. Factors which could increase the risk of ischaemic stroke include transient or chronic hypertension [20, 21], decreased blood flow in middle cerebral artery [22–25]; increased blood viscosity [26] and impaired fibrinolysis [27]; decreased venodilatory responsiveness to bradykinin [28]; impaired hypercapnic ventilatory response [29], elevated sympathetic tone [30], increased epinephrine-related platelet activation [31]; increased disposition to atherosclerosis [32, 33], and decreased vascular reactivity [34]. The strong relationship between OSAS and hypertension is well documented [36–39] and it is well known that hypertension is the most important risk factor for haemorrhagic stroke [7, 40]. In most sleep apnoea studies, haemorrhagic and ischaemic stroke patients were included and analysed together. Since in our view a different mechanism of cerebrovascular lesion necessarily implicates also a different relationship to sleep apnoeas and OSAS, we wanted to analyse ischaemic and haemorrhagic stroke patients distinctly. In this study we followed a cohort of acute ischaemic and haemorrhagic stroke patients for sleep apnoeas. Patients and Methods106 patients (73 ischaemic strokes, mean age 67 B 14 years; 45 males and 28 females and 33 haemorrhagic strokes, mean age 60 B 11 years; 25 males and 8 females) were included and screened for sleep apnoeas in 6 days after stroke (test 1). The stroke diagnosis was substantiated by clinical data and neuroimaging (CT or MRI) per- formed within 48 h after admission. Neuroimaging was repeated within 6 weeks in all patients and those with secondary haemorrhagic transformation of ischaemic infarction were excluded. From the initial population, 51 persons (37 ischaemic; mean age 64 ± 11 years; 26 men, 11 women, and 14 haemorrhagic mean age 61 ± 10 years; 11 men and 3 women) with pathological oxygen desaturation index (ODI > 10) were included in the follow-up study. Sleep apnoea screening was repeated 3 months after the first record (test 2). Sleep apnoea assessment was performed by MESAM IV, a portable device developed for home sleep-apnoea screening [41, 42]. We evaluated the main sleeping period, 4 h between midnight and 4 a.m., unless personal observation and/or manual analysis of the MESAM IV records revealed that the sleeping period of a patient occurred at a different time of the night. Then we analysed the MESAM IV record at that period (table 1).
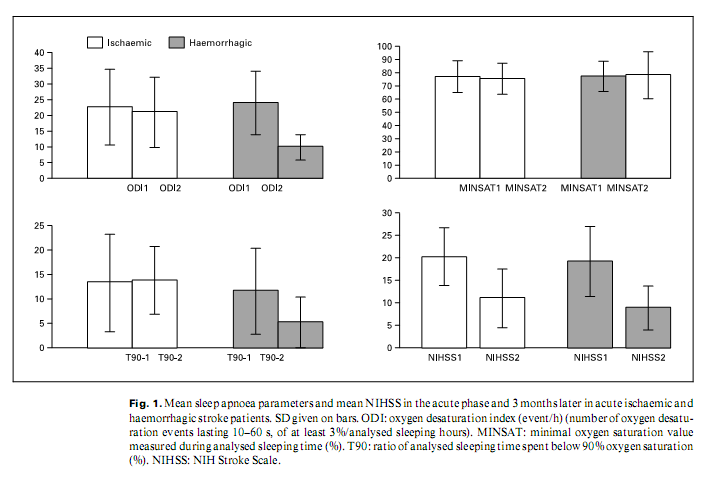
O2 saturation values (sampling in every 2 s), snoring sound, heart rate (sampling in every second both) and body position were recorded. From the available parameters ODI: number of desaturation events of at least 3%/h; ratio of sleeping time spent below 90% haemoglobin saturation (T90), and minimal oxyhaemoglobin saturation value during sleeping time (MINSAT) were chosen to characterize breathing during sleep. All patients had a detailed medical history including risk-profile assessment. NIH stroke scale (NIHSS) was used to quantify neurological deficits at the time of the first and second sleep analysis. Snoring history was gathered from the patients or bed partners. Statistical Evaluation To assess the effects of stroke parameters and risk factors observed at test 1 on sleep apnoea parameters and NIHSS at test 2, general linear models were developed. In each model one of the four outcome measures served as dependent variable (i.e. change in DESAT, MINSAT, T90, and NIHSS). Localization of stroke (brainstem-cerebellar vs. other), stroke type (haemorrhagic or ischaemic), gender, age and risk factors as hypertension, ResultsTen patients of the initial acute stroke population (9%) died in the observational period, 7 of them had an ODI > 20/h. In the ischaemic stroke group, loud snoring before stroke was more frequent than in the haemorrhagic group (44% of men and 25% of women in the ischaemic group, 20% of men and 0% of women in the haemorrhagic group). Risk profiles of the ischaemic and haemorrhagic patients’ group were similar, except for hypercholesterolaemia, being more frequent in the ischaemic population (p = 0.002). Risk profiles of patients with pathological (ODI > 10/h) versus those with normal (ODI ≤ 10) sleep apnoea frequency were different in the ischaemic group. Those with pathological apnoea frequency snored more often before stroke (p = 0.002); there were more patients with arterial hypertension (p = 0.002) than in the nonapnoeic group. In the haemorrhagic group there was no important difference between those with pathological and ‘normal’ sleep apnoea frequency. Seventy percent of the acute ischaemic and 64% of the acute haemorrhagic stroke group had a sleep apnoea frequency characterized by ODI > l0; 45% of the ischaemic and 40% of the haemorrhagic stroke patients had a sleep apnoea frequency with an ODI > 20. Localization of stroke had no effect on sleep apnoea frequency in the acute phase. In the follow-up group improvement of NIHSS was similar in the ischaemic and haemorrhagic groups, however, changing of sleep apnoea parameters (ODI, T90, and MINSAT) was different: ODI of ischaemic stroke patients did not change, but there was a significant improvement of ODI in the haemorrhagic group (p = 0.0002) (fig. 1). Also the MINSAT and T90 values improved less in ischaemic than in haemorrhagic stroke (NS). There was no correlation between the improvement of sleep apnoea parameters and the change of NIHSS in ischaemic stroke, but there was a tendency of correlation between them in haemorrhagic stroke. MINSAT improved best (p = 0.0001) in posterior circulation haemorrhagic strokes.
There was no significant difference in the acute phase (test 1) in NIHSS values in those with normal versus those with pathologic sleep apnoea frequency, and there was no negative correlation between ODI measured in test 1 and the change of NIHSS on follow-up. DiscussionIn this study we registered pathological sleep apnoea frequency in 64–70% of acute stroke patients. Similar data were also found in other studies [12–17]. This apnoea frequency greatly exceeds prevalence values of OSAS in any age group of the general population [1–3, 43]. Three months after the acute stroke, in spite of the global clinical improvement, sleep apnoea frequency and severity were unchanged in most ischaemic stroke patients. Loud snoring, a strong indicator for OSAS [6, 44, 45], was frequent before stroke in this group. Sleep apnoea frequency and severity greatly improved in haemorrhagic strokes, tending to move along with clinical improvement; loud snoring was infrequent before stroke. We can speculate that the two major stroke types have a different relationship to pathological sleep apnoea frequency. Most cerebral bleeding is likely to cause ‘de novo’ sleep apnoeas, improving along with other symptoms. On the other hand, high sleep apnoea frequency would be a concomitant phenomenon in most ischaemic strokes that pre-existed to the cerebrovascular event, remaining unchanged, while clinical stroke symptoms improve. A recent study [46] found that central apnoeas improve on 3 months’ follow-up after acute ischaemic and haemorrhagic strokes while obstructive apnoeas remain stable. Interpreting those results together with ours we are presuming that haemorrhagic strokes lead more often to central apnoeas, and ischaemic strokes are characteristically accompanied by OSAS as a probable risk factor [5–9]. Our data suggest that for patients with pathological sleep apnoea frequency detected in the acute phase of ischaemic stroke, long-term treatment for OSAS will be necessary. In contrast, sleep apnoeas in acute haemorrhagic stroke may temporarily need special care, but would probably improve along with global clinical improvement. Our follow-up tendencies did not confirm those experiences [13] that the functional outcome after stroke is less favourable in patients having higher ODI. On the other hand, high frequency of severe sleep apnoea among those deceased after test 1, suggests an increased vulnerability in the pathological sleep apnoea group. The small study population and the great number of factors taken into account is one of the main limitations of this study. To avoid erroneous interpretations, we set the critical p value to as low as 0.0005. However, sparse data in this field induced us to report also tendencies. The MESAM IV system is a widely used tool for sleep apnoea screening. It has been is validated with polysomnography. According to these tests it has a sensitivity of 92% and a specificity of 97% in detecting sleep apnoeas [47–51]. The disadvantage of the system is that breathing events – hypopnoeas, obstructive and central apnoeas – cannot be reliably discriminated; traditional polysomnography with breathing parameters would be more informative. References1 Partinen M, Telakivi T: Epidemiology of obstructive sleep apnea syndrome. Sleep 1992; 15(6 suppl):S1–S4. 2 Redline S, Young T: Epidemiology and natural history of obstructive sleep apnea. Ear Nose Throat J 1993;72:20–21. 3 Bixler EO, Vgontzas AN, Ten Have T, Tyson K, Kales A: Effects of age on sleep apnea in men. I. Prevalence and severity. Am J Respir Crit Care Med 1998;157:144–148. 4 Partinen M, Guilleminault C: Daytime sleepiness and vascular morbidity at seven-year follow-up in obstructive sleep apnoea patients. Chest 1990;97:27–32. 5 Palomäki H: Snoring and the risk of ischemic brain infarction. Stroke 1991;22:1021–1025. 6 Fletcher EC: Obstructive sleep apnoea and cardiovascular morbidity. Monaldi Arch Chest Dis 1996;51:77–80. 7 Weiss JW, Launois SH, Anand A, Garpestad E: Cardiovascular morbidity in obstructive sleep apnoea. Progr Cardiovasc Dis 1999;41:367– 376. 8 Shepard JW Jr: Hypertension, cardiac arrhythmias, myocardial infarction, and stroke in relation to obstructive sleep apnoea. Clin Chest Med 1992;13:437–458. 9 Neau JP, Meurice JC, Paquereau J, Chavagnat JJ, Ingrand P, Gil R: Habitual snoring as a risk factor for brain infarction. Acta Neurol Scand 1995;92:63–68. 10 Pressman MR, Schetman WR, Figueroa WG, Van Uitert B, Caplan HJ, Peterson DD: Transient ischemic attacks and minor stroke during sleep. Relationship to obstructive sleep apnoea syndrome. Stroke 1995;26:2361–2365. 1 12 Dyken ME, Somers VK, Yamada T, Ren ZY, Zimmerman, B: Investigating the relationship between stroke and obstructive sleep apnoea. Stroke 1996;27:401–407. 13 Good DC, Henkle JQ, Gelber D, Welsh J, Verhulst S: Sleep-disordered breathing and poor functional outcome after stroke. Stroke 1996; 27:252–259. 14 Bassetti C, Aldrich MS, Quint D: Sleep-disordered breathing in patients with acute supra- and infratentorial strokes. A prospective study of 39 patients. Stroke 1997;28:1765–1772. 15 Bassetti C, Aldrich MS: Sleep apnoea in acute cerebrovascular diseases: Final report on 128 patients. Sleep 1999;22:217–223. 16 Wessendorf TE, Teschler H, Wang YM, Konietzko N, Thilmann AF: Sleep-disordered breathing among patients with first-ever stroke. J Neurol 2000;247:41–47. 17 Askenasy JJ, Goldhammer I: Sleep apnoea as a feature of bulbar stroke. Stroke 1988;19:637– 639. 18 Hudgel DW, Devadatta P, Quadri M, Sioson ER, Hamilton H: Mechanism of sleep-induced periodic breathing in convalescing stroke patients and healthy elderly subjects. Chest 1993; 104:1503–1510. 19 Mohsenin V, Valor R: Sleep apnoea in patients with hemispheric stroke. Arch Phys Med Rehabil 1995;76:71–76. 20 Fletcher EC: The relationship between systemic hypertension and obstructive sleep apnea: Facts and theory. Am J Med 1995;98:118– 128. 21 Klingelhöfer J, Hajak G, Sander D, Schulz- Varszegi M, Rüther E, Conrad B: Assessment of intracranial hemodynamics in sleep apnoea syndrome. Stroke 1992;23:1427–1433. 22 Weiss JW, Remsburg S, Garpestad E, Ringler J, Sparrow D, Parker JA: Hemodynamic consequences of obstructive sleep apnoea. Sleep 1996;19:388–397. 23 Siebler M, Nachtmann A: Cerebral hemodynamics in obstructive sleep apnoea. Chest 1993;103:1118–1119. 24 Netzer N, Werner P, Jochums I, Lehmann M, Strohl KP: Blood flow of the middle cerebral artery with sleep-disordered breathing: Correlation with obstructive hypopneas. Stroke 1998;29:87–93. 25 Droste DW, Ludemann P, Anders F, Kemeny V, Thomas M, Krauss JK, Ringelstein EB: Middle cerebral artery blood flow velocity, end-tidal pCO2 and blood pressure in patients with obstructive sleep apnoea and in healthy subjects during continuous positive airway pressure breathing. Neurol Res 1999;21:737– 741. 26 Nobili L, Schiavi G, Bozano E, de Carli F, Ferrillo F, Nobili F: Morning increase of whole blood viscosity in obstructive sleep apnoea syndrome. Clin Hemorheol Microcirc 2000;22: 21–27. 27 Hedner J, Calson J, Rangenmark C, Gleerup G, Winther K: Platelet function and fibrinolytic activity in patients with sleep apnoea. J Sleep Res 1994;3(suppl l):101. 28 Duchna HW, Guilleminault C, Stoohs RA, Faul JL, Moreno H, Hoffman BB, Blaschke TF: Vascular reactivity in obstructive sleep apnoea syndrome. Am J Respir Crit Care Med 2000;161:187–191. 29 Qureshi AI, Christopher Winter W, Bliwise DL: Sleep fragmentation and morning cerebrovasomotor reactivity to hypercapnia. Am J Respir Crit Care Med 1999;160:1244–1247. 30 Waradekar NV, Sinoway LI, Zwillich CW, Leuenberger UA: Influence of treatment on muscle sympathetic nerve activity in sleep apnoea. Am J Respir Crit Care Med 1996;153: 1333–1338. 31 Eisensehr I, Ehrenberg BL, Noachtar S, Korbett K, Byrne A, McAuley A, Palabrica: Platelet activation, epinephrine, and blood pressure in obstructive sleep apnoea syndrome. Neurology 1998;51:188–195. 32 Aboyans V, Lacroix P, Virot P, Tapie P, Cassat C, Rambaud G, Laskar M, Bensaid J: Sleep apnoea syndrome and the extent of atherosclerotic lesions in middle-aged men with myocardial infarction. Int Angiol 1999;18:70–73. 33 Friedlander AH, Friedlander IK, Yueh R, Littner MR: The prevalence of carotid atheromas seen on panoramic radiographs of patients with obstructive sleep apnoea and their relation to risk factors for atherosclerosis. J Oral Maxillofac Surg 1999;57:516–521. 34 Placidi F, Diomedi M, Cupini LM, Bernardi G, Silvestrini M: Impairment of daytime cerebrovascular reactivity in patients with obstructive sleep apnoea syndrome. J Sleep Res 1998; 7:288–292. 35 Young T, Peppard P, Palta M, Hla KM, Finn L, Morgan B, Skatrud J: Population-based study of sleep-disordered breathing as a risk factor for hypertension. Arch Intern Med 1997; 157:1746–1752. 36 Working Group on OSA and Hypertension: Obstructive sleep apnoea and blood pressure elevation: what is the relationship? Blood Press 1993;2:166–182. 37 Zwillich CW: Obstructive sleep apnoea causes transient and sustained systemic hypertension. Int J Clin Pract 1999;53:301–305. 38 Lavie P, Herer P, Hoffstein V: Obstructive sleep apnoea syndrome as a risk factor for hypertension: Population study. BMJ 2000; 320:479–482. 39 Wessendorf TE, Teschler H, Wang YM, Sorgenfrei U, Thilmann AF: Obstruktives Schlaf- apnoesyndrom wahrscheinliche Ursache einer therapieresistenten Hypertonie bei intrazerebraler Blutung. Nervenarzt 1999;70:927–930. 40 Ancoli-Israel S, Kripke DF, Mason W Partinen M, Telakivi T: Characteristics of obstructive and central sleep apnea in the elderly: An interim report. Biol Psychiatry 1987;22:741–750. 41 Rihs F, Mathis J, Gugger M, Hess CD: Screening für Schlafapnoe: Klinische Anwendung von Mesam IV und Apnoe-Check. Schweiz Med Wochenschr 1995;995–1002. 42 Rauscher H, Popp W, Zwick H: Quantification of sleep disordered breathing by computerized analysis of oxymetry, heart rate and snoring. Eur Respir J 1991;4:655–659. 43 Bresnitz EA, Goldberg R, Kosinski RM: Epidemiology of obstructive sleep apnea. Epidemiol Rev 1994;16:210–227. 44 Philip P, Dealberto MJ, Dartigues JF, Guilleminault C, Bioulac B: Prevalence and correlates of nocturnal desaturations in a sample of elderly people. J Sleep Res 1997;6:264–271. 45 Puvanendran K, Goh KL: From snoring to sleep apnoea in a Singapore population. Sleep Res Online 1999;2:11–14. http://www.sro.org/ 1999/Puvanendran 46 Parra O, Arboix A, Bechich S, Garcıá-Eroles L, Montserrat JM, López JA, Ballester E, Guerra JM, Sopeña JJ: Time course of sleep-related breathing disorders in first-ever stroke or transient ischemic attack. Am J Respir Crit Care Med 2000;161:375–380. 47 Esnaola S, Durán J, Infante Rivard C, Rubio R, Fernández A: Diagnostic accuracy of a portable recording device (MESAM IV) in suspected obstructive sleep apnoea. Eur Respir J 1996;9: 2597–2605. 48 Stoohs R, Guilleminault C: MESAM 4: An ambulatory device for the detection of patients at risk for obstructive sleep apnoea syndrome. Chest 1992;101:1221–1227. 49 Verse T, Pirsig W, Junge Hülsing B, Kroker B: Validierung einer 7kanaligen ambulanten Polygraphieeinheit. 2. Simultane Polysomnographie. HNO 1999;47:256–261. 50 Fletcher EC, Goodnight White S, Munafo D, Miller CC 3rd, Luckett R, Qian W: Rate of oxyhemoglobin desaturation in obstructive versus non-obstructive apnea. Am Rev Respir Dis 1991;143:657–660. 51Schäfer H, Ewig S, Hasper E, Lüderitz B: Predictive diagnostic value of clinical assessment and non-laboratory monitoring system recordings in patients with symptoms suggestive of obstructive sleep apnea syndrome. Respiration 1997;64:194–199.
|