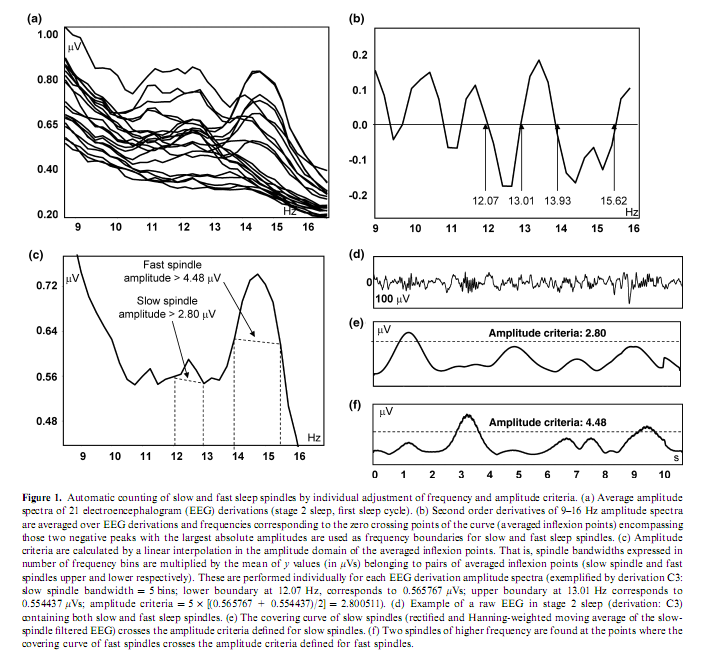
Authors:RÓBERT BÓDIZS1, TAMÁS KIS2, ALPÁR SÁNDOR LÁZÁR1, LINDA HAVRÁN3, PÉTER RIGÓ1, ZSÓFIA CLEMENS4 and PÉTER HALÁSZ4 1 Institute of Behavioural Sciences, Semmelweis University, Budapest, Hungary Correspondence: Róbert Bódizs, Institute of Behavioural Sciences, Semmelweis University, Accepted in revised form 10 June 2005; received 23 September 2004 SUMMARYThe usual assessment of general mental ability (or intelligence) is based on performance attained in reasoning and problem-solving tasks. Differences in general mental ability have been associated with event-related neural activity patterns of the wakeful working brain or physical, chemical and electrical brain features measured during wakeful resting conditions. Recent evidences suggest that specific sleep electroencephalogram oscillations are related to wakeful cognitive performances. Our aim is to reveal the relationship between non-rapid eye movement sleep-specific oscillations (the slow oscillation, delta activity, slow and fast sleep spindle density, the grouping of slow and fast sleep spindles) and general mental ability assessed by the Raven Progressive Matrices Test (RPMT). The grouping of fast sleep spindles by the cortical slow oscillation in the left frontopolar derivation (Fp1) as well as the density of fast sleep spindles over the right frontal area (Fp2, F4), correlated positively with general mental ability. Data from those selected electrodes that showed the high correlations with general mental ability explained almost 70% of interindividual variance in RPMT scores. Results suggest that individual differences in general mental ability are reflected in fast sleep spindle-related oscillatory activity measured over the frontal cortex. keywords: cortical synchronization, electroencephalogram, intelligence, mental test, prefrontal cortex, sleep spindles INTRODUCTIONNetwork oscillations are essential parts of brain functioning (Buzsáki and Draguhn, 2004). The cortical slow (<1 Hz) oscillation, the thalamocortically and cortically generated delta activity (1–4 Hz) as well as sleep spindling (12–15 Hz) emerging from thalamo-cortico-thalamic loops are characteristic forms of non-rapid eye movement (NREM) sleep-specific oscillations (Steriade, 2003). Some forms of sleep-dependent network oscillations reciprocally interact with synaptic plasticity (Miyamoto and Hensch, 2003). This was proven for sleep spindle oscillations, which promote plasticity phenomena like cortical augmenting responses (Steriade, 2003; Steriade and Timofeev, 2003) and the triggering of hippocampal discharges related to memory consolidation (Sirota et al., 2003). On the contrary, sleep itself is a plastic process affected by waking experiences. Sleep spindles and sleep-electroencephalogram (EEG) delta waves are enhanced by verbal and motor learning respectively (Gais et al., 2002; Huber et al., 2004). There is a region-specific decrease in sleep-EEG delta activity in the visual cortex of cats and mice as a consequence of the reduced visual input in the dark-rearing condition (Miyamoto et al., 2003). Both phasic sleep spindle events and the sleep-EEG power spectra have been shown to have high internight reliability (Gaillard and Blois, 1981; Silverstein and Levy, 1976; Tan et al., 2000). The rate of occurrence of sleep spindles can vary between 3 and 8 per min in different individuals, but is very stable across two nights in the same subject (Gaillard and Blois, 1981). Other results demonstrate that the pattern of the EEG power distribution in NREM sleep is characteristic for an individual, possibly, reflecting individual traits of functional anatomy (Finelli et al., 2001). The stability of some sleep-EEG measures is also suggested by their correlations with relatively stable cognitive abilities. Preliminary reports suggest positive correlations between the number of night-time sleep spindles and full scale as well as performance IQ (Nader and Smith, 2001). Measures of the slow oscillation were shown to be associated with visuospatial memory (Bódizs et al., 2002) and performance at tasks more specific to the left prefrontal cortex (Anderson and Horne, 2003). Although spindle oscillations emerge from thalamo-cortico-thalamic loops (Steriade, 2003), they reflect intracortical synaptic activity (Kandel and Buzsáki, 1997) being effectively triggered, grouped and shaped by the cortical slow (<1 Hz) oscillation (Steriade, 2003), which is initiated in layer 5 (Sanchez-Vives and McCormick, 2000) and synchronized by cortico-cortical synapses (Amzica and Steriade, 1995; Steriade, 2003). Sleep spindling has been shown to be maximal in the surface-positive phases of the slow oscillation, which expresses its triggering and grouping by the synchronized cortical activity of <1 Hz (Contreras and Steriade, 1996; Fell et al., 2002; Mölle et al., 2002; Steriade, 2003). These data suggest that cortical network efficacy manifests itself in the power and density of the slow oscillation and sleep spindles respectively. In humans, two types of spindles have been described: slow spindles with a frequency of about 12 Hz and predominant frontal localization and fast spindles of about 14 Hz with predominant, but not exclusive centroparietal localization (Anderer et al., 2001; De Gennaro and Ferrara, 2003; Zeitlhofer et al., 1997). There are controversies regarding the physiological basis and functional significance of these two types of sleep spindles. The neural network efficiency theory of intelligence considers that general mental ability is a function of a general feature of the brain, probably brain connectivity (Anderson, 1995). Based on the above-mentioned electrophysiological studies and in accordance with the neural network efficiency theory of intelligence (Anderson, 1995) we hypothesize that individual traits of the above-mentioned sleep EEG oscillations (slow oscillation, delta waves, slow and fast sleep spindles) and the grouping of slow and fast sleep spindles by the slow oscillation, which depend on neural connectivity (Amzica and Steriade, 1995) predict general mental ability. In contrast with the study of Gais et al. (2002) showing the spindle enhancement in the first part of the night after daytime verbal learning, we designed an experiment in which differences in daytime activity were minimized. Therefore, possible differences in the previously mentioned sleep EEG parameters could be attributed mainly to differences in subjects baseline EEG characteristi METHODSSubjects and procedures In order to test our hypothesis 19 healthy subjects (14 males, five females), with elementary or high-school degree were recruited by newspaper advertisement. In order to get a better estimate of the culture-free general factor in mental abilities no subjects with university degree were selected for this study. Eighteen of the subjects were aged between 27 and 47 years, and one subject was 67 years of age. Neither the exclusion of the latter subject nor the exclusion of the five female subjects modified the results of the study. The research protocol was approved by the Ethical Committee of the National Institute of Psychiatry and Neurology. All subjects signed informed consent for the participation in the study. They slept two consecutive nights in the sleep laboratory, according to their preferred sleep schedules. Sleep was monitored by standard polysomnography including 21 EEG derivations of the 10–20 system (Fp1, Fp2, Fpz, F3, F4, Fz, F7, F8, C3, C4, Cz, T3, T4, T5, T6, P3, P4, Pz, O1, O2, Oz) referred to the contralateral mastoids, as well as bipolar electro-oculography, submental electromyography and electrocardiography with collodion-fixed Ag/AgCl electrodes. We used the right mastoid as a reference for central EEG derivations. However, the Cz electrode was referred to both the right and the left mastoid and these channels were used in the validation procedure of our spindle detection algorithm (see below). Signals from all electrodes were cut-off filtered at 0.33 Hz, amplified and digitized at 128 Hz with 12-bit resolution. Subjects spent the day between the two consecutive recording nights in the department completing psychometric tests at fixed time schedules. Intelligence was assessed with the standard form of the Raven Progressive Matrices Test (RPMT) (Raven et al., 1976) between 10 : 00 and 11 : 00 hours. The RPMT is a paper-and-pencil measure of abstract reasoning. The test consists of five sets of 12 different 3 x 3 matrices of geometric patterns, gradually increasing in difficulty. The bottom right pattern of each matrix was missing. For each matrix, six alternatives to fill in the missing pattern are presented and the participants are asked to identify the piece required to complete the design. The total number of correct items was used in statistical analyses. Statistical evidence suggests that performance on this reasoning task is the best measure of the general factor in mental abilities (Gray and Thompson, 2004). Outside the standard testing, subjects were restricted in doing major physical and mental effort in order to avoid large interindividual differences in daytime activity and its consecutive effects on sleep. Although one cannot directly test for mental efforts, our subjects could only choose either reading (newspapers or their own books) or watching TV (three local channels). Physical activity was restricted by the electrodes that were attached to the subjects head during the whole study. No daytime naps were allowed and subjects were instructed to refrain from drinking alcohol or taking any medication. EEG analysis Sleep recordings of the second nights were visually scored according to standard criteria (Rechtschaffen and Kales, 1968). After visual artefact rejection, 4-s epochs were Hanning-tapered and fast Fourier transformed in order to get the average amplitude spectra for stage 2 sleep and slow-wave sleep (stages 3 and 4) with 0.25-Hz resolution. Absolute and relative amplitude spectra of the slow (0.5–1.25 Hz) and delta (1.5–4.5 Hz) activity were calculated for slow-wave sleep by the integration of the 0.5–1.25 and 1.5–4.5 Hz spectral curves and by their division with the integrated total amplitude spectra (for relative amplitude spectra). Slow and fast spindles were counted by an automatic method described below (see also Fig. 1). Based on the theory that slow spindles could be in fact anterior peaks of alpha activity (De Gennaro and Ferrara, 2003) individual slow and fast sleep spindle frequencies were determined in the 9–16 Hz frequency range, which covers both alpha and sigma bands. The second order derivatives of 9–16 Hz amplitude spectra of stage 2 sleep EEGs were averaged over EEG derivations and frequencies corresponding to the zero crossing points (averaged inflexion points) encompassing those two negative peaks with the largest absolute amplitudes were defined as frequency criteria for slow and fast sleep spindles. Spindles were defined as waves with above-mentioned frequency and with amplitudes falling in the corresponding peak region (over the line determined by the average inflexion points) of the average amplitude spectra. Hence amplitude criteria for slow and fast spindles were calculated from the average amplitude spectra by linear interpolation in the amplitude domain of the averaged inflexion points (lower and upper frequency criteria) of slow and fast spindles respectively. This was performed individually for amplitude spectra of each EEG derivation, allowing us to control the distance between the active electrode and the reference affecting amplitude measures (inflexion points of the amplitude spectra vary with the amplitude measure of the EEG). Slow or fast spindles were counted if the covering curve of the rectified slow or fast spindle-filtered EEGs exceeded the slow or fast spindle amplitude criteria respectively. Filtering was performed by a third order Butterworth bandpass filter followed by reverse filtering in order to avoid phase distortion. The boundaries of the filter were set to 2 x 0.1 Hz tighter than the boundaries calculated by the technique mentioned above in order to finally obtain a better match between visual and automatic spindle detection. The covering curve of the filtered and rectified signals was determined by 11 points Hanning-weighted moving averages multiplied by 4.41 in order to compensate for amplitude attenuation during averaging.
Slow and fast spindle densities of individual NREM–REM cycles were calculated by dividing the number of slow and fast spindles by minutes of stage 2 sleep during which they occurred. All-night spindle densities were obtained by averaging the data of individual NREM–REM cycles. The robustness of the spindle detection algorithm described above was validated by the comparison of stage 2 sleep spindle densities obtained in the first NREM–REM cycle for Cz-right mastoid and Cz-left mastoid channels. Our spindle detection algorithm resulted in almost perfect correlations between the sleep spindle densities of the right and left mastoid referred Cz channels. They were r = 0.95 (P = 5 x 10-10 ) and r = 0.98 (P = 2 x 10-13) for slow and fast sleep spindle densities, respectively, suggesting that the choice of reference electrode was unlikely to have affected the results. In order to establish the above-mentioned algorithm’s accuracy in the identification of visually detected sleep spindles, up to 20 min of stage 2 sleep EEG from the first sleep cycle was analysed separately with respect to derivation Fz. Two experienced raters (RB and ASL) scored independently the sleep spindles in this EEG channel. Criteria for sleep spindles were the following: a group of waves with 11–15 Hz frequency (covering the usual range of both slow and fast sleep spindles in most of the subjects), lasting at least 0.5 s (maximum 2 s) and having a more or less spindle-like shape. The two ratings were unified and disagreements solved by consensus. Results of the automatic slow and fast sleep spindle detection procedures were also unified on this EEG segments. These unified automatic procedures correctly identified 90.13% (between 79% and 100%) of the visually scored sleep spindles. Quantification of the grouping of sleep spindles by the slow oscillation (<1 Hz) was performed by calculating slow half- wave triggered averages in spi Partial correlations between RPMT scores and EEG data (absolute and relative amplitude spectra of the slow oscillation and delta waves, respectively, as well as slow and fast spindle density) were calculated with the statistical control of age. This was performed for all-night slow-wave sleep (slow oscillation and delta waves) and stage 2 sleep (slow and fast sleep spindling). Partial correlations between RPMT and grouping of spindles by the slow oscillation were calculated for all-night slow-wave sleep (stage 3 and 4). In order to obtain a better localization of regions with significant correlations between EEG and RPMT results of the above analyses were represented by significance probability maps (Hassainia et al., 1994). As multiple testing can inflate type I error, the sequentially rejective Bonferroni approach was applied (Holm, 1979). This reduces the risk of type I error at the cost of increasing the risk of type II error, but give information about the relationship between specific variables (specific EEG derivations and RPMT scores). RESULTS
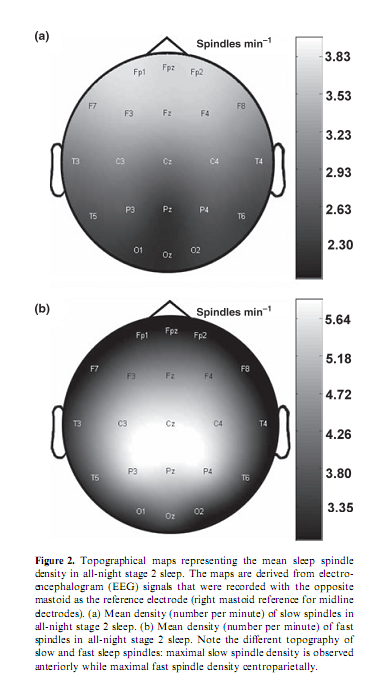
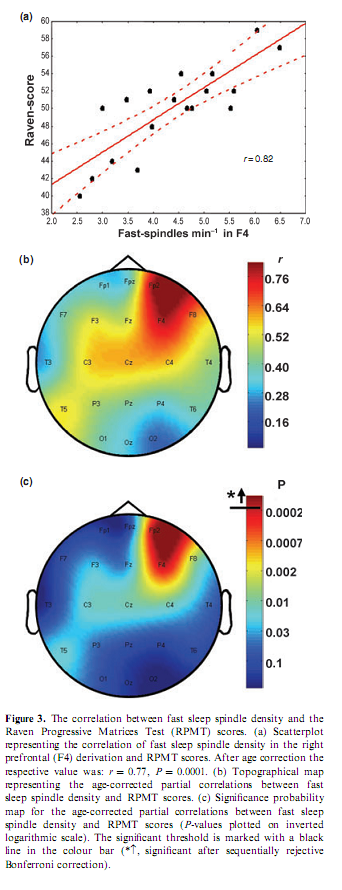
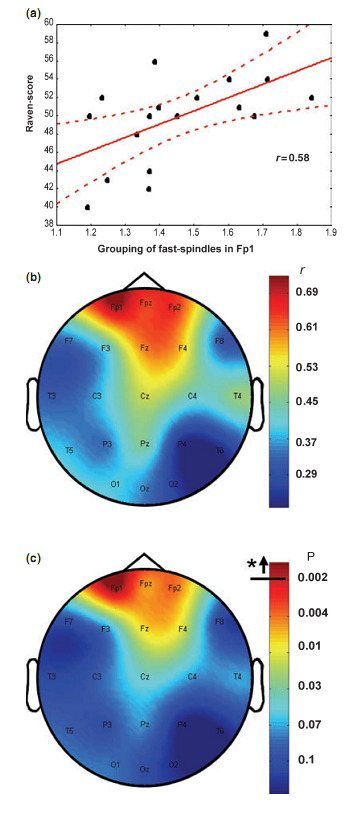
Percentage of time spent in stage 2 sleep moderately correlated with RPMT scores (r = 0.49; P = 0.034). Absolute and relative amplitude spectra of the slow oscillation (0.50– 1.25 Hz) or delta waves (1.5–4.5 Hz) during stages 3 and 4 sleep did not correlate with test scores. There was a different topographical distribution of slow and fast sleep spindling: slow spindles predominated anteriorly, while fast sleep spindles centroparietally (Fig. 2). The mean lower and upper limits of the frequency ranges for slow spindles were 11.175 Hz (range 10.092–12.060 Hz, SD = 0.588) and 12.102 Hz (range 10.744–13.057 Hz, SD = 0.623) respectively. Mean lower and upper limits for fast spindles were 13.114 Hz (range 12.046–14.145 Hz, SD = 0.577) and 14.380 Hz (range 13.088–15.390 Hz, SD = 0.634) respectively. Slow sleep spindles did not correlate with RPMT scores (data not shown). In contrast, fast spindle density correlated positively with RPMT scores. The density of fast spindles recorded from right prefrontal (F4) and frontopolar (Fp2) derivations correlated positively with RPMT scores after sequentially rejective Bonferroni correction (F4: r = 0.77, P = 0.000179; Fp2: r = 0.79, P = 0.000126; see also Fig. 3). Maximal spindling always occurred in the positive phases or after approximately 400 ms of the negative phases of the slow oscillation, but the relative peak-to-through values showed substantial interindividual and regional variation. The grouping of slow spindles did not correlate with RPMT scores in any area. In contrast, the grouping of fast spindles correlated positively with RPMT scores. Grouping of fast spindles recorded from the left frontopolar derivation (Fp1) correlated positively with RPMT scores after Bonferroni correction (Fp1: r = 0.70, P = 0.0014; see also Fig. 4). Two strong sleep-EEG correlates of general mental ability (fast spindle density in derivation F4 and grouping of fast spindles in derivation Fp1) explained almost 70% of the interindividual variance in RPMT scores (R2 = 0.69, F = 18.545, d.f. = 2, 16, P < 0.00007).
DISCUSSIONHitherto intelligence differences have been associated with event-related neural activity patterns of the wakeful working brain (Gray et al., 2003) or physical (Andreasen et al., 1993; Thompson et al., 2001), chemical (Jung et al., 1999; Rae et al., 1996) and electrical (Jausovec and Jausovec, 2000) brain features measured during wakeful resting conditions. Our results show that general mental ability reliably correlates with fast spindle density as well as with the grouping of fast spindles by the slow oscillation. These findings cohere with the preliminary report of a positive correlation between the total number of sleep spindles for the night and performance IQ and full scale IQ respectively (Nader and Smith, 2001). As the sleep-EEG of the prefrontal derivations showed the most robust correlations with RPMT scores in our study, present results are in accordance with data supporting the role of the prefrontal cortex in intelligent behaviour (Duncan et al., 2000; Gray and Thompson, 2004; Gray et al., 2003) as well as with the relationship between prefrontal grey matter volume and IQ (Posthuma et al., 2002; Thompson et al., 2001). The deterioration of intellectual functions is an inherent property of dementia and demented patients were shown to have a reduced number of sleep spindles when compared with non- demented, age-matched controls (Petit et al., 2004). Hitherto changes in the topographical distribution of sleep spindles of demented patients have not been analysed, but we would hypothesize an association between the reduction in frontal fast spindles and cognitive impairments assessed by the RPMT. Although our results are in line with other reports showing a predominant anterior localization of slow spindles (Anderer et al., 2001; De Gennaro and Ferrara, 2003; Zeitlhofer et al., 1997), there was a lack of association between slow spindles and RPMT scores. This might relate to their different physiological source as suggested by the predominance of frontal slow spindles during the period of immaturity of frontal lobes (Shinomiya et al., 1999). Moreover, there is evidence suggesting that slow spindles could be in fact anterior peaks of alpha activity during NREM sleep (De Gennaro and Ferrara, 2003). Despite the fact that fast spindles were more abundant in centroparietal derivations in our study and in other reports as well (Anderer et al., 2001; De Gennaro and Ferrara, 2003; Zeitlhofer et al., 1997), subjects characterized by relatively high frontal fast spindle density and grouping were shown to perform better on the RPMT. Thus, effective frontal triggering and grouping of fast spindles during NREM sleep correlates with general mental ability. Recently, Anderson and Horne (2003) reported significant associations between 0.5 and 1.0 Hz power from the left frontal EEG channel in the first NREM period and performance at tasks more specific to the left prefrontal cortex in healthy older people. A high level of slow (0.5–1.0 Hz) oscillatory activity during sleep was associated with a better performance in prefrontal cortical tasks. However, negative correlations between 0.5 and 1.0 Hz power from the right frontal EEG channel in the first NREM period, and fluid intelligence measured by the Cattell test. As there is strong evidence that the lateral prefrontal cortex supports intelligent behaviour (Duncan et al., 2000; Gray and Thompson, 2004; Gray et al., 2003), their results are somewhat inconsistent in this regard. We did not find significant correlations between flu As the daytime activity of our subjects was controlled in this study, correlations reflect a relationship between baseline characteristics of individual-specific sleep EEG features and general mental ability. The density and grouping of sleep spindles, which are triggered by the slow oscillation (Steriade, 2003) depending on intracortical connectivity (Amzica and Steriade, 1995) correlate with general mental ability measured by the RPMT. Thus, general mental ability is related to intracortical connectivity of neural networks. This result is in accordance with the hypothesis of Anderson (1995) that brain connectivity is responsible for the biological correlates of IQ discovered so far. This does not mean that learning has no effect on corticothalamic and neocortico-hippocampal processes during night- time sleep. The latter hypothesis is supported by the study of Gais et al. (2002). However, there are some important issues to rise in this regard. The possibility exists that slow and fast sleep spindles are differentially involved in these processes. Despite the fact that there is evidence for the functional differentiation of slow and fast sleep spindles (De Gennaro and Ferrara, 2003; Shinomiya et al., 1999), these were not differentially measured in the Gais et al. (2002) study. Finally, learning performance correlated positively with baseline (prelearning) sleep spindle density in the Gais et al. (2002) report, which suggests a trait-like relationship between sleep and cognition similar to that observed in the present study. However, one cannot rule out the possibility that use dependency of spindles could be reflected throughout the night. In motor learning, final stages of off-line stage 2 sleep- dependent neuronal reprocessing and consolidation of memory traces are hypothesized to be related to sleep spindling in the second half of the night (Smith et al., 2004). Taking into consideration the above-mentioned theory and the fact that high IQ persons perform better on motor learning demanding their non-dominant hands than low IQ persons (Tan, 1989), the possibility exists that differences in spindle density of our subjects reflect the brain correlate of some motor learning processes. Several studies suggest that sleep spindles have a sleep protective function playing an active role in inducing and maintaining sleep and affecting arousal by blocking the transmission of external stimuli through the thalamus to the cortex (De Gennaro and Ferrara, 2003). This could mean that general mental ability correlates not only with sleep spindle density, but also with a less disturbed sleep. However, there was quite weak evidence supporting this hypothesis in our results. Although percentage of time spent in stage 2 sleep correlated positively with performance in the RPMT, no relationship between RPMT scores and signs of disturbed sleep, like wake after sleep onset or stage 1 sleep was observed. Therefore, we agree with the view that to the extent to which sleep spindles can be considered to play a role in sleep preservation by inhibiting or attenuating potentially arousing stimuli, these effects appear to be restricted to endogenously generated stimuli and are passive rather than reactive in nature (Pivik et al., 1999). In conclusion, the above results suggest that cortical network efficiency, particularly in prefrontal areas, ensuring intelligent behaviour during wakefulness is reflected in effective triggering and grouping of fast sleep spindles during night-time NREM sleep. According to the effect size estimates for Pearson correlation coefficients, correlations above 0.50 are of crucial practical importance (Hojat and Xu, 2004). Therefore, our results raise the possibility of the prominent significance of sleep-EEG in unravelling the neural bases of human cognitive functions. ACKNOWLEDGEMENTSThe authors wish to thank Zoltán Somogyvári for his helpful advices concerning data analysis. This work was supported by grants of the Hungarian Medical Research Council (ETT-162/2003) and the National Office for Research and Technology (NKFP-1B/020/04). REFERENCESAmzica, F. and Steriade, M. Disconnection of intracortical synaptic linkages disrupts synchronization of a slow oscillation. J. Neurosci., 1995, 15: 4658–4677. Anderer, P., Klosch, G., Gruber, G., Trenker, E., Pascual-Marqui, R. D., Zeitlhofer, J., Barbanoj, M. J., Rappelsberger, P. and Saletu, B. Low-resolution brain electromagnetic tomography revealed simultaneously active frontal and parietal sleep spindle sources in the human cortex. Neuroscience, 2001, 103: 581–592. Anderson, B. G explained. Med. Hypotheses, 1995, 45: 602–604. Anderson, C. and Horne, J. A. Prefrontal cortex: links between low frequency delta EEG in sleep and neuropsychological performance in healthy, older people. Psychophysiology, 2003, 40: 349–357. Andreasen, N. C., Flaum, M., Swayze, V., II, O’Leary, D. S., Alliger, R., Cohen, G., Ehrhardt, J. and Yuh, W. T. Intelligence and brain structure in normal individuals. Am. J. Psychiatry, 1993, 150: 130– 134. Bódizs, R., Békésy, M., Szűcs, A., Barsi, P. and Halász, P. Sleep- dependent hippocampal slow activity correlates with waking memory performance in humans. Neurobiol. Learn. Mem., 2002, 78: 441– 457. Buzsáki, G. and Draguhn, A. Neuronal oscillations in cortical networks. Science, 2004, 304: 1926–1929. Contreras, D. and Steriade, M. Spindle oscillation in cats: the role of corticothalamic feedback in a thalamically generated rhythm. J. Physiol. (Lond.), 1996, 490 (Suppl. 1): 159–179. De Gennaro, L. and Ferrara, M. Sleep spindles: an overview. Sleep Med. Rev., 2003, 7: 423–440. Duncan, J., Seitz, R. J., Kolodny, J., Bor, D., Herzog, H., Ahmed, A., Newell, F. N. and Emslie, H. A neural basis for general intelligence. Science, 2000, 289: 457–460. Fell, J., Elfadil, H., Röschke, J., Burr, W., Klaver, P., Elger, C. E. and Fernández, G. Human scalp recorded sigma activity is modulated by slow EEG oscillations during deep sleep. Int. J. Neurosci., 2002, 112: 893–900. Finelli, L. A., Achermann, P. and Borbély, A. A. Individual fingerprints in human sleep EEG topography. Neuropsychophar- macology, 2001, 25: S57–S62. Gaillard, J.-M. and Blois, R. Spind Gais, S., Mölle, M., Helms, K. and Born, J. Learning-dependent increases in sleep spindle density. J. Neurosci., 2002, 22: 6830–6834. Gray, J. R. and Thompson, P.M. Neurobiology of intelligence: science and ethics. Nat. Rev. Neurosci., 2004, 5: 471–482. Gray, J. R., Chabris, C. F. and Braver, T. S. Neural mechanisms of general fluid intelligence. Nat. Neurosci., 2003, 6: 316–322. Hassainia, F., Petit, D. and Montplaisir, J. Significance probability mapping: the final touch in t-statistic mapping. Brain Topogr., 1994, 7: 3–8. Hojat, M. and Xu, G. A visitor’s guide to effect sizes: statistical significance versus practical (clinical) importance of research findings. Adv. Health Sci. Educ., 2004, 9: 241–249. Holm, S. A simple sequentially rejective multiple test procedure. Scand. J. Stat., 1979, 6: 65–70. Huber, R., Ghilardi, M. F., Massimini,M. and Tononi, G. Local sleep and learning. Nature, 2004, 430: 78–81. Jausovec, N. and Jausovec, K. Differences in resting EEG related to ability. Brain Topogr., 2000, 12: 229–240. Jung, R. E., Brooks, W. M., Yeo, R. A., Chiulli, S. J., Weers, D. C. and Sibbitt, W. L., Jr. Biochemical markers of intelligence: a proton MR spectroscopy study of normal human brain. Proc. R. Soc. Lond., B, 1999, 266: 1375–1379. Kandel, A. and Buzsáki, G. Cellular–synaptic generation of sleep spindles, spike-and-wave-discharges, and evoked thalamocortical responses in the neocortex of the rat. J. Neurosci., 1997, 17: 6783– 6797. Miyamoto, H. and Hensch, T. K. Reciprocal interaction of sleep and synaptic plasticity. Mol. Interv., 2003, 3: 404–417. Miyamoto, H., Katagiri, H. and Hensch, T. Experience-dependent slow-wave sleep development. Nat. Neurosci., 2003, 6: 553–554. Mölle, M., Marshall, L., Gais, S. and Born, J. Grouping of spindle activity during slow oscillations in human non-rapid eye movement sleep. J. Neurosci., 2002, 22: 10941–10947. Nader, R. and Smith, C. Intelligence and stage 2 sleep spindles. Actas Fisiol., 2001, 7: 125. Petit, D., Gagnon, J. F., Fantini, M. L., Ferini-Strambi, L. and Montplaisir, J. Sleep and quantitative EEG in neurodegenerative disorders. J. Psychosom. Res., 2004, 56: 487–496. Pivik, R. T., Joncas, S. and Busby, K. A. Sleep spindles and arousal: the effects of age and sensory stimulation. Sleep Res. Online, 1999, 2: 89–100 (Available at: http://www.sro.org/1999/Pivik/89/). Posthuma, D., De Geus, E. J., Baare, W. F., Hulshoff Pol, H. E., Kahn, R. S. and Boomsma, D. I. The association between brain volume and intelligence is of genetic origin. Nat. Neurosci., 2002, 5: 83–84. Rae, C., Scott, R. B., Thompson, C. H., Kemp, G. J., Dumughn, I., Styles, P., Tracey, I. and Radda, G. K. Is pH a biochemical marker of IQ? Proc. R. Soc. Lond., B, Biol. Sci., 1996, 263: 1061–1064. Raven, J. C., Court, J. H. and Raven, J. Manual for Raven’s Progressive Matrices. H. K. Lewis, London, 1976. Rechtschaffen, A. and Kales, A. Manual of Standardized Terminology, Techniques and Scoring System for Sleep Stages of Human Subjects. UCLA Brain Information Service/Brain Research Institute, Los Angeles, 1968. Sanchez-Vives, M. V. and McCormick, D. A. Cellular and network mechanisms of rhythmic recurrent activity in neocortex. Nat. Neurosci., 2000, 3: 1027–1034. Shinomiya, S., Nagata, K., Takahashi, K. and Masumura, T. Development of sleep spindles in young children and adolescents. Clin. Electroencephalogr., 1999, 30: 39–43. Silverstein, L. D. and Levy, C. M. The stability of the sigma sleep spindle. Electroencephalogr. Clin. Neurophysiol., 1976, 40: 666– 670. Sirota, A., Csicsvári, J., Buhl, D. and Buzsáki, G. Communication between neocortex and hippocampus during sleep in rodents. Proc. Natl. Acad. Sci. U.S.A., 2003, 100: 2065–2069. Smith, C. T., Aubrey, J. B. and Peters K. R. Different roles for REM and stage 2 sleep in motor learning: a proposed model. Psychol. Belg., 2004, 44: 81–104. Steriade, M. The corticothalamic system in sleep. Front. Biosci., 2003, 8: d878–d899. Steriade, M. and Timofeev, I. Neuronal plasticity in thalamocortical networks during sleep and waking oscillations. Neuron, 2003, 37: 563–576. Tan, U. Right and left hand skill in left-handers: distribution, learning, and relation to nonverbal intelligence. Int. J. Neurosci., 1989, 44: 235–249. Tan, X., Campbell, I. G., Palagini, L. and Feinberg, I. High internight reliability of computer-measured NREM delta, sigma, and beta: biological implications. Biol. Psychiatry, 2000, 48: 1010–1019. Thompson, P. M., Cannon, T. D., Narr, K. L., van Erp, T., Poutanen, V. P., Huttunen, M., Lonnqvist, J., Standertskjold-Nordenstam, C. G., Kaprio, J., Khaledy,M., Dail, R., Zoumalan, C. I. and Toga, A. W. Genetic influences on brain structure. Nat. Neurosci., 2001, 4: 1253–1258. Zeitlhofer, J., Gruber, G., Anderer, P., Asenbaum, S., Schimicek, P. and Saletu, B. Topographic distribution of sleep spindles in young healthy subjects. J. Sleep Res., 1997, 6: 149–155.
|